Human immunoglobulin is an immunological drug. It is a concentrated solution of an immunologically active protein fraction, which is isolated from the blood plasma of healthy donors by fractionation with ethyl alcohol at temperatures below 0°C.
To produce one batch of immunoglobulin, manufacturers use plasma obtained from at least 1000 healthy donors. They are preliminarily individually tested for the absence of surface antigen of the hepatitis B virus, antibodies to the hepatitis C virus and human immunodeficiency viruses.
At the Yusupov Hospital, immunoglobulins registered in the Russian Federation are used to treat patients. They are highly effective and have a minimal range of side effects.
Instructions for use
The active ingredients of normal human immunoglobulin are immunoglobulins, which contain antibodies of various specificities. The preparation contains from 9.5 to 10.5% protein. The maximum concentration of antibodies in the blood is determined 24-48 hours after administration of the drug. The half-life of antibodies is 4-5 weeks.
Normal human immunoglobulin (instructions are in the box) is available in the form of a solution in ampoules of 1.5 ml (1 dose). One package may contain 5, 10 or 20 ampoules of the drug. The kit includes an ampoule nail file. The drug is dispensed in pharmacies with a doctor's prescription. Immunoglobulin in ampoules is transported and stored at air temperatures from +2 to +8°C.
Immunoglobulin is injected intramuscularly into the outer upper quadrant of the buttock or the anterior surface of the thigh. The drug is not administered intravenously. Nurses at the Yusupov Hospital strictly follow the rules of asepsis and antisepsis when performing immunoglobulin injections. Before injection, ampoules with human immunoglobulin are kept for two hours at room temperature.
To prevent foam from forming in the syringe, the drug is drawn into the syringe with a wide-bore needle. It is injected by changing the needle. The drug in an opened ampoule cannot be stored. At the Yusupov Hospital, patients are not administered immunoglobulin if the integrity or labeling of the ampoules is damaged. The drug is unsuitable for use if the solution becomes cloudy, changes color, has flakes that do not break, as well as immunoglobulin that was stored in improper conditions or has expired.
Make an appointment
Indications
Immunoglobulin is not used as a vaccine or medicine. It is designed to strengthen the body's defenses in the fight against diseases, as well as to develop a lasting barrier against them. Immunoglobulins contain antibodies that recognize harmful bacteria and viruses that enter the body, inhibit their reproduction and neutralize the toxic substances they produce.
Doctors at the Yusupov Hospital use normal human immunoglobulin to prevent various diseases:
- for immunodeficiency, immune diseases;
- in the postoperative period;
- for the prevention of diseases such as influenza, whooping cough, measles, meningococcal infection, polio, in addition, the drug has an antitetanus effect;
- for the prevention of tick-borne encephalitis;
- for various infectious diseases, including sepsis;
- for blood diseases;
- with acquired immunodeficiency syndrome (usually children), etc.
Doctors at the Yusupov Hospital prescribe immunoglobulin 25 ml for severe forms of bacterial-toxic and viral infections, sepsis, and dermatomyositis. The drug is included in the treatment regimen for Guillain-Barré syndrome, hyperimmunoglobulinemia E syndrome, Eaton-Lambert syndrome, and infections caused by parvovirus B9.
Neurologists prescribe simple human immunoglobulin to patients suffering from multiple sclerosis, chronic inflammatory demyelination in polyneuropathy, and myasthenia gravis. Pediatricians use the drug to prevent and treat infections in newborns, premature babies and children born with low birth weight.
After the introduction of normal human immunoglobulin, the overall resistance of the body increases during the recovery period of patients with infectious diseases.
Pharmacological properties of the drug Human immunoglobulin
Immunobiological agent, highly purified polyvalent human immunoglobulin. Immunoglobulin contains about 90% monomeric IgG and a small fraction of decomposition products, dimeric and polymeric IgG and IgA, IgM in trace concentrations. The distribution of IgG subclasses in it corresponds to their fractional distribution in human serum. It has a wide range of opsonizing and neutralizing antibodies against bacteria, viruses and other pathogens. In patients with primary or secondary immunodeficiency syndromes, it provides replenishment of missing IgG class antibodies, which reduces the risk of developing infection. In some other immune disorders, such as idiopathic (immune origin) thrombocytopenic purpura and Kawasaki syndrome, the mechanism of clinical effectiveness of immunoglobulin is not entirely clear. After IV infusion, a redistribution of immunoglobulin occurs between the blood plasma and the extravascular space, and equilibrium is achieved after approximately 7 days. Antibodies present in exogenous immunoglobulin have the same pharmacokinetic characteristics as antibodies in endogenous IgG. In individuals with normal serum IgG levels, the biological half-life of immunoglobulin averages 21 days, while in patients with primary hypogammaglobulinemia or agammaglobulinemia, the half-life of total IgG averages 32 days (however, there is significant individual variation that may be important when establishing a dosage regimen for a particular patient).
Contraindications
Normal human immunoglobulin should not be used by persons suffering from the following pathologies:
- hypersensitivity to human immunoglobulins;
- renal failure;
- diabetes mellitus;
- during exacerbation of allergic processes, etc.
Contraindications to the use of human immunoglobulin are severe allergic reactions to the administration of blood products in the past (allergic rashes, Quincke's edema, anaphylactic shock). The drug should not be used by patients suffering from systemic immunopathological diseases - connective tissue diseases, blood pathologies, nephritis. The use of simple immunoglobulin is contraindicated in thrombocytopenia and other disorders of the blood coagulation system.
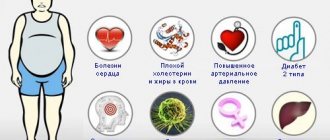
Human immunoglobulin is used with caution in people with obesity, diabetes mellitus, and in the presence of risk factors for thrombotic complications (arterial hypertension, pathologies of the cardiovascular system, increased blood viscosity, old age). The drug is prescribed strictly under the supervision of a doctor in case of pathologies of the urinary system.
With extreme caution, normal human immunoglobulin is used for pregnant and lactating women, as well as for persons with decompensated chronic heart failure and suffering from migraines.
Analogs
Level 4 ATC code matches:
Pentaglobin
Octagam
A number of analogues of this drug are used in medicine. These are the drugs Gamunex , Immunovenin , Intraglobin , Pentaglobin , Intratect , Gabriglobin , Endobulin , etc.
To choose an adequate analogue, you should understand what Immunoglobulin is and be sure to consult a doctor. To decide which drug will be most optimal for the treatment or prevention of diseases, for example, Immunoglobulin or Yodantipyrine for the prevention of tick-borne encephalitis , you should definitely consult with a specialist.
Side effects
Provided that all recommendations for dosage, administration and precautions are followed, serious side effects when using human normal immunoglobulin develop in extremely rare cases. Symptoms may appear several hours or even days after using the drug. As a rule, after stopping the use of immunoglobulin, side effects disappear.
Most often, side effects occur at a high rate of administration of the drug. By reducing the speed and temporarily stopping the intake, you can eliminate the majority of side effects. In other situations, symptomatic therapy is carried out.
The maximum likelihood of side effects occurs during the first use of the drug, during the first 60 minutes. Patients may experience the development of a flu-like syndrome with malaise, chills, fever, weakness, and headache.
In addition, the development of the following symptoms may be observed in various body systems:
- from the respiratory system: the appearance of a dry cough, shortness of breath;
- from the gastrointestinal tract: the appearance of nausea, diarrhea, vomiting, stomach pain, increased salivation;
- from the cardiovascular system: development of tachycardia, cyanosis, chest pain, flushing of the face;
- from the central nervous system (weakness, drowsiness, symptoms of aseptic meningitis (rarely) - nausea, vomiting, headache, photosensitivity, impaired consciousness, stiffness of the neck muscles);
- from the renal system: development of tubular necrosis, worsening renal failure in patients with renal dysfunction.
In addition to all of the above, when taking normal human immunoglobulin, the development of allergic reactions (skin rashes, bronchospasm, itching) and local reactions (hyperemia at the injection site) may occur.
It is extremely rare that when using the drug, loss of consciousness, collapse, or a sharp increase in blood pressure to significant levels may occur. In such situations, the drug must be discontinued and the patient must be immediately given antihistamines, adrenaline, and plasma replacement solutions.
After administration of human immunoglobulin, pain in the muscles of the limbs or back, numbness, fever, or a feeling of cold sometimes occurs. The skin at the injection site may turn red.
Make an appointment
Use of the drug during pregnancy
The role of immunoglobulins is especially important in the treatment of diseases in pregnant women. Their body is extremely sensitive to medications, many of them are simply contraindicated. Immunoglobulins are simply irreplaceable in this situation. They are non-toxic and do not harm the body. These are the drugs that increase the level of a person’s defense system and help him independently overcome the disease or prevent its development.
However, studies on the reaction of the body of pregnant women to the action of the drug have not been conducted, so there is no information about how harmful or harmless normal human immunoglobulin is during pregnancy and lactation. During pregnancy, the drug can be used in situations where there is an urgent need for its administration, if the potential benefits to the expectant mother's body outweigh the possible risks to the fetus.
During breastfeeding, normal human immunoglobulin should also be used with extreme caution, as it can enter the baby’s body through breast milk.
What is immunoglobulin (IG)
Immunoglobulins or antibodies are protein compounds in the blood plasma that are produced as a response to the entry of various antigens (bacteria, viruses, toxins, etc.) into the body. Antibodies prevent the proliferation of foreign organisms and block their production of toxins. Immunoglobulins are an important part of the human immune system. They are used as identifiers of harmful foreign objects, causing an immune response. There are five classes of immunoglobulins, each of which has a specific composition and differs in function:
- IgG;
- IgA;
- IgM;
- IgD;
- IgE.
In case of deficiency or incapacity of immunoglobulins, their administration from the outside is prescribed. The intravenous route of administration is often used. Immunoglobulins are obtained from donor blood plasma from more than 1000 donors. The immunologically active protein fraction is necessarily checked for the absence of antibodies to primary and secondary immunodeficiency, hepatitis C and B viruses.
For intravenous administration, class G immunoglobulins are predominantly used. The drug replenishes natural antibodies in the blood serum. It has immunomodulatory and immunostimulating effects. Immunoglobulins are effective in immunodeficiency states and in the treatment of severe diseases of a viral and bacteriological nature.
Drug interactions
Human normal immunoglobulin is considered pharmaceutically incompatible with other drugs. A separate dropper or syringe must be used for its administration. The simultaneous use of this drug with active immunization agents (for rubella, measles, chickenpox and mumps) may cause a decrease in the effectiveness of therapy. Live viral vaccines administered parenterally can be used no earlier than one month after the administration of normal human immunoglobulin.
In addition, in order to avoid the development of negative consequences, simultaneous administration of this drug with calcium gluconate to infants should not be allowed.
Conditions for dispensing from pharmacies
Normal human immunoglobulin (25 ml) is available by prescription. The decision to use normal human immunoglobulin should be made by a qualified specialist, based on the results of the examination and the characteristics of the patient’s body.
Independent use of the drug for other purposes may lead to negative consequences. You cannot buy immunoglobulin at non-specialized points of sale (from hand, according to an advertisement, with a large discount or a very low price), since in this case the storage conditions of the drug are often violated or the drug is not an immunoglobulin.
Immunoglobulin price, where to buy
The price of normal human immunoglobulin for intramuscular administration is 850-900 rubles. for 10 ampoules. Where to buy Human Immunoglobulin in Moscow, Yekaterinburg and other cities, where to get an injection, and how much Human Immunoglobulin costs, you can find out at your nearest medical facility. The price of Immunoglobulin IV is on average 2600 rubles per 25 ml bottle. The cost of an injection (ampoule) in Russia (Moscow, Chelyabinsk, etc.) is about 100 rubles.
- Online pharmacies in RussiaRussia
- Online pharmacies in UkraineUkraine
ZdravCity
- Human immunoglobulin anti-rhesus rho (d) solution for intramuscular administration 300 µg/dose 1 ml ampoule 1 pcs Ivanovo Regional Blood Transfusion Station
RUR 5,251 order
show more
Special instructions for the use of human immunoglobulin
Due to the fact that in case of individual intolerance to the drug, patients may develop an immediate type of allergy, including anaphylactic shock, after the administration of simple human immunoglobulin, patients are under the supervision of a doctor at the Yusupov Hospital for 30 minutes. The drug is used with caution in case of impaired renal function. For children, pediatricians individually select the dose of the drug.
Since immunoglobulin reduces the effectiveness of vaccination, vaccinations are carried out no earlier than 2-3 months after administration of the drug. A temporary increase in antibodies in the blood after the administration of immunoglobulin leads to false-positive analysis data in a serological study. Pregnant women are administered simple human immunoglobulin only when the expected benefit to the mother outweighs the potential risk to the fetus. Immunoglobulin passes into breast milk and helps prevent infectious diseases in newborns.
It is recommended to observe the speed of administration, since faster administration may cause the development of collapse. The drug should be stored and transported at a temperature of +2 to +8°C. Immunoglobulin ampoules should not be frozen.
IMMUNOGLOBULINS for intravenous administration in pediatrics
What is the criterion for the effectiveness of immunomodulators? What are the indications for the use of IVIG?
The rapid development of fundamental and applied immunology is accompanied by the intensive introduction of immunocorrection methods into healthcare practice. Currently, doctors quite widely use immunomodulators of various pharmacological groups and mechanisms of action.
Immunomodulation in the broad sense of the term implies an effect on various parts of the immune system, leading to an increase or decrease in the parameters of the immune response in order to maintain or restore immune homeostasis. It has now been established that hundreds of drugs of various classes have an immunocorrective effect. Moreover, drugs that affect certain parts of the immune system are not only special substances, but also drugs that have long been widely used in practical medicine, such as vitamins, amino acids, enzymes, adaptogens, endocrine agents, some cardiovascular and nootropic drugs drugs, etc.
Despite the long experience of using immunomodulators, assessing their effectiveness is one of the most difficult issues in modern medicine. And first of all, because changes in immune status are extremely labile over time and individual, and many of the identified deviations do not so much reflect a dysfunction of the immune system, but rather indicate an adaptive response of the body to pathological influences. In addition, we must not forget that we still cannot really identify the entire spectrum of existing breakdowns of the body’s immune response. That is why today the criteria for the effectiveness of immunomodulators are medical and statistical indicators, such as morbidity and mortality rates, the incidence of complications and transition to chronic pathology, recovery time, etc.
Among the numerous types of immunomodulatory therapy, immunoreplacement therapy should be noted, which includes the use of immunoglobulin preparations, fresh frozen plasma, leukemia concentrate, recombinant colony-stimulating factor (CSF) preparations, interleukins, interferons, thymic factors, etc.
Of these drugs, intravenous immunoglobulins (IVIGs) occupy a prominent place, especially in the treatment of children, although many issues related to their use remain unresolved. It must be emphasized: the very widespread opinion among doctors that the main area of use of IVIg is severe infection does not at all correspond to the therapeutic capabilities of these drugs.
As is known, immunoglobulins for intravenous administration replaced immunoglobulins for intramuscular administration, in which the dose of the administered drug varied significantly depending on the ratio of the rate of absorption and its inactivation at the injection site. Currently, immunoglobulins for intramuscular administration have practically lost their importance.
IVIg was originally proposed for the treatment of primary immunodeficiencies, for which their effectiveness has been proven. Diseases such as X-linked agamma- and hypogammaglobulinemia, common variable hypogammaglobulinemia, immunodeficiency with hypergammaglobulinemia M, Wiskott-Aldrich, Louis-Bart syndromes, X-linked lymphoproliferative syndrome, severe combined immunodeficiencies of all types are direct indications for therapy with intravenous immunoglobulins. Along with this, the effectiveness of IVIg has been proven for transient hypogammaglobulinemia in children, with Kawasaki disease, as well as for the prevention of cytomegalovirus infection during bone marrow transplantation [2].
Since the advent of sandoglobulin, which contains an intact molecule with active Fc function and has an immunomodulating effect, in the early 1980s, the scope of use of IVIg has expanded significantly [2]. Currently, the therapeutic effect of IVIG has been proven in acute and chronic immune thrombocytopenia and immune neutropenia, as well as in chronic demyelinating inflammatory polyneuropathy. It is worth noting that high doses of drugs are used for immune pathology.
A large number of studies indicate the high effectiveness of IVIg in the treatment of severe bacterial infections, including in young children and neonatal sepsis [4]. It is also proposed to use immunoglobulins in high-risk newborns to prevent the development of infection [4], however, convincing data on the preventive effectiveness of this method of therapy have not yet been obtained.
The use of IVIG in the treatment of severe bacterial and viral-bacterial infections in children has a convincing pathogenetic justification. It is known that during severe, especially generalized, infection, an excess of products of normal or impaired metabolism, cellular reaction products, is formed in the patient’s body. These substances, accumulating, become endogenous toxic substances for the body, causing intoxication. The presence of both bacterial antigenemia and toxemia in a patient aggravates intoxication and leads to disruption of normal intercellular interactions in the immune response. This situation intensifies with the development of multiple organ failure, which accompanies severe infection and sepsis. And in newborns and children in the first six months of life, in addition, there is physiological hypogammaglobulinemia, which also helps to strengthen this mechanism.
Intercellular interactions occurring during the immune response play a key role in its adequacy, and, consequently, in the possibility of recovery. Intense intoxication during an infectious disease is accompanied by a decrease in the content of antigen-binding lymphocytes. The affinity of their receptors also decreases due to their blocking and suppression of expression by inflammatory mediators, that is, in fact, depression of the cellular component of immunity is observed.
In addition, infectious toxicosis is, to one degree or another, accompanied by an increase in the level of R-protein in plasma, a product of the catabolism of cellular receptors, which has ligand-binding and immunoregulatory properties. Increased concentrations of R proteins also contribute to immunosuppression. Moreover, it was noted that their level correlates with the severity of the disease and the severity of metabolic changes in the body.
Drug therapy, including antibacterial therapy, for severe metabolic disorders can provide only a minimal effect, since the effectiveness of patient treatment ultimately, even with the absolutely correct selection of drugs, depends on the ability of the body’s cells to respond to the therapy. IVIg, by binding and eliminating many components that cause intoxication, helps unblock receptors and normalize antigen expression. These drugs also help restore the ability of phagocytes and effectors of natural cytotoxicity (natural killer cells, T-lymphocytes, etc.) to carry out anti-infective protection, increase the number of corresponding receptors of various cell populations capable of responding adequately to the influence of bacterial inducers, hormones, interferons and other biologically active substances. In other words, IVIg helps increase the sensitivity of immunocompetent cells of the macroorganism to drug therapy and increases anti-infective protection.
Thus, to achieve maximum effectiveness of anti-infective therapy for severe infection and sepsis, it is most advisable to combine antibacterial therapy with intravenous immunoglobulin [5, 9]. Moreover, if we are talking about children, then, given the presence of a tendency towards hypogammaglobulinemia in newborns, and especially in premature newborns, and young children, it is advisable to administer IVIg until the level of Ig G in the blood reaches at least 800 mg%. The recommended daily dose is from 500 to 800 mg/kg of body weight, and the duration of the course of administration is from 3 to 6 days.
Administration of immunoglobulin should be carried out as early as possible after diagnosis of an infectious disease and in sufficient volume. Studies conducted at our department [1] showed that the administration of standard immunoglobulins (i.e., immunoglobulins with a predominant Ig G content, such as intraglobin F, etc.) to newborn children in the first week of a severe infectious disease (meningitis, sepsis) was accompanied by improvement in 57% of cases. Moreover, with a total dose of IVIG within 5-6 ml/kg of body weight, the effect was observed only in 33-40% of observations. If the total dose of the drug reached 9-10 ml/kg of body weight, a positive effect was recorded in 70% of cases. However, a subsequent increase in the total dose to 15-18 mg/kg body weight was not accompanied by a significant increase in effectiveness. Prescribing IVIg at a later date - at 3-5 weeks of illness - was also ineffective (34-32%). This may be due to the fact that increased doses of intravenously administered human Ig G preparations have the ability to inhibit the production of interleukins and reduce the level of expression of IL-2 receptors due to the presence in the preparations of antibodies against the corresponding interleukins and blockade of Fc receptors [6]. In addition, IVIGs contain a number of biologically active proteins, for example, soluble HLA class I and II molecules, which are also important for the implementation of the immune response.
As noted above, the immunomodulatory effect of IVIG is closely related to the presence of the Fc function of the immunoglobulin molecule, as well as the content of Ig G subpopulations (primarily the Ig G2 subpopulation, the synthesis of which is reduced in children of the first year of life) and Ig M. Preservation of the Fc function , in turn, largely depends on the methods used to inactivate viruses in the preparation and can vary significantly in different immunoglobulin preparations.
Currently, there are about 10 IVIG drugs from various manufacturers registered in Russia. First of all, it should be noted that among all IVIGs, it is customary to distinguish the most extensive group of so-called standard polyvalent immunoglobulins, which include all preparations of normal donor immunoglobulin G: sandoglobulin, alphaglobin, endobulin, intraglobin F, octagam, domestic immunoglobulin for intravenous administration, etc. Their mechanism of action and clinical effect are approximately the same. However, drugs differ in their virus safety, ability to cause adverse reactions, and requirements for their storage and use.
The first standard IVIGs, or first generation standard IVIGs, were treated with alkylation, causing them to lose the Fc moiety and have little or no immunomodulatory effect. Therefore, these IVIGs were used purely for replacement purposes in cases of immunodeficiency. Currently, they are outdated and have rather historical significance.
In the late 70s of the last century, sandoglobulin was created, called a second-generation drug. Its fundamental difference was the presence of an intact immunoglobulin molecule with preserved active Fc function (70-75% of the European standard). It was the emergence of this IVIG that made it possible to significantly expand the indications for the clinical use of immunoglobulins.
Third generation drugs (eg, intraglobin) were developed in the 80s. They are characterized by a high degree of purification (5 stages of purification), high virus safety, and almost 100% activity of the Fc fragment. The drugs are available in liquid form and are stored in the refrigerator. Immunoglobulins of this class are characterized by a high clinical effect in the treatment of severe bacterial infections, especially those caused by gram-positive microflora, as well as in the treatment of immune pathologies.
Immunoglobulins, which appeared in the 90s (in particular, octagam, alphaglobin), also have a high degree of purity (4 stages of purification), virus safety and Fc-fragment activity. The ratio of immunoglobulin subclasses in them is characterized by a higher content of the Ig G3 subclass, that is, the subclass of virus-neutralizing antibodies. Hence the high effectiveness of these IVIGs not only for bacterial infections, but also for generalized viral (for example, enterovirus) and viral-bacterial diseases (for example, Kawasaki disease). At the same time, the use of this IVIg does not provide convincing effectiveness against generalized cytomegalovirus infection [3]. The advantages of Octagam include the fact that this immunoglobulin can be stored at room temperature, so the drug can be used as soon as the need arises.
Standard immunoglobulins also differ in osmolarity level and pH value. These parameters to some extent determine the possibility of side effects and should be taken into account when administering large doses of the drug to children with multiple organ failure and homeostasis disorders. Thus, drugs that are hyperosmolar in relation to blood plasma are more likely to cause renal dysfunction, which should be taken into account when prescribing them. Drugs with low pH affect the acid-base balance of the body. Among the widely used IVIGs, sandoglobulin has the highest osmolarity, and alphaglobin and octagam have the lowest pH (5.4-6.0).
The second group of IVIGs consists of drugs enriched with Ig M. But this group of immunoglobulins is represented in Russia by one drug - pentaglobin (Biotest Pharma, Germany). It contains 12% Ig M, or 6 g/l. In other IVIG preparations, the Ig M content does not exceed 0.1 g/l. An exception is Venimmun N immunoglobulin (USA), where the Ig M content reaches 5% [10], but this drug is not registered in Russia.
The presence in the preparation of class M immunoglobulin, which is the first to be formed in response to antigenic irritation and carries antibodies to endotoxin and capsular antigens of gram-negative bacteria, made pentaglobin extremely effective in the treatment of severe infectious diseases. In addition, Ig M antibodies fix complement better than other classes of Ig and accordingly improve opsonization, that is, the preparation of bacteria for phagocytosis. It should be especially emphasized that pentaglobin significantly increases the opsonization of E. coli, K. pneumoniae, Ps. aeruginosa, S. pneumoniae, H. influenzae. It is noted that an increase in the level of Ig M in the body helps to limit production, increase inhibition of pro-inflammatory cytokines and, accordingly, limit the general inflammatory response of the body, which underlies the development of multiple organ failure in infectious-toxic shock and sepsis.
Intravenous administration of pentaglobin was accompanied by a significantly significant increase in Ig M levels on days 3-5 after administration [5, 9]. A meta-analysis of 20 clinical studies of the use of pentaglobin in the treatment of neonatal sepsis showed that the use of the drug significantly (more than 2 times) reduces mortality from this disease, while the administration of standard immunoglobulins leads to a reduction in mortality by only 1.37 times [11].
Finally, a special group consists of immunoglobulins enriched with specific antibodies. Examples of such drugs are cytotect, which is characterized by an increased titer of anti-cytomegalovirus antibodies, and hepatect, which contains antibodies to the hepatitis B virus.
The use of Cytotect is indicated for the prevention and treatment of cytomegalovirus infection, for example, during tissue and organ transplantation, drug immunosuppression, and secondary immunodeficiency. For neonatal CMV infection, this is the only drug for antiviral therapy, since the use of ganciclovir is possible only at an older age. A dose of 50 IU/kg body weight per day is usually used. The frequency of administration is from 3 to 6 times, depending on the clinical picture and condition of the patient.
Gepatect is an IVIG drug enriched with virus-neutralizing antibodies to the hepatitis B virus. The drug is used for active-passive immunization (simultaneous use of hepatitis B vaccine and immunoglobulin in newborns), as well as independently, when due to the severity of the condition it is impossible to vaccinate a child born from the mother - carriers of the hepatitis A virus. Active-passive immunization using hepatect is carried out only for those children who were born from mothers with current acute hepatitis, from mothers who are early convalescents of hepatitis and Hbs Ag carriers. The usual dose is 20 IU/kg of the child’s weight [12].
The overall main advantage of using intravenous immunoglobulin preparations is the ability to quickly and easily achieve high serum concentrations of immunoglobulins. Data have been obtained on the high degree of safety of intravenous administration of conventional therapeutic doses of immunoglobulins for infectious diseases in newborns; Thus, the use of IVIG in premature infants in doses up to 1000 mg/kg per day did not have any side effects [7, 8]. But the expansion of the use of IVIG, especially the increase in doses practiced to achieve an immunomodulatory effect, has made it possible to discover a number of new side effects and undesirable effects of the drugs.
Previously, the main side effects of IVIG were associated with non-compliance with the requirements for the speed of administration - too fast administration. This was usually manifested by pain, nausea, vomiting, fever, chills and changes in blood pressure.
With an increase in the dose of IVIG, a side effect such as reversible acute renal failure was observed, caused by plasma hyperosmolarity created during the administration of the drug. Typically, acute renal failure occurs when immunoglobulin is used in concentrations higher than 3% and 5% solutions.
The occurrence of aseptic meningitis has also been described, the development mechanism of which is associated with the effect of human leukocyte antigen molecules and the action of cytokines. The influence of specific autoantibodies cannot be excluded. Aseptic meningitis has nothing in common with infectious meningitis and requires only purely symptomatic treatment.
The use of ultra-high doses of IVIG (for immune pathology) can be accompanied by hemolysis and disseminated intravascular coagulation; cases of hemolytic anemia have also been described. These complications are associated with a conflict in the ABO system (the first two) and the presence of anti-D-isoagglutinins (hemolytic anemia). Therefore, when using ultra-high doses, it is recommended to check IVIG for blood group compatibility and the presence of anti-D-agglutinins.
It should be noted that in most cases, side effects during IVIG therapy are mild and completely reversible. However, despite this, a strictly substantiated approach to their purpose is required.
Literature
- Alekseeva V.V. Infections of the central nervous system with a protracted course in children of the first year of life (Clinical and immunological characteristics and immunocorrection): Diss. ...cand. honey. Sci. M., 2000.
- Martin T.D. //Therapeutic archive. 1996. T. 68. No. 10.
- Samsygina G. A., Dudina T. A. // Pediatrics. 2001. No. 3. P. 83-86.
- Tabolin V. A., Volodin N. N., Degtyareva M. V. et al. Immunocorrection for bacterial infections in newborns: reality and prospects.
- Abraham E., Glauser MP, Butler T. et al. // JAMA. 1997. 277 (19): 1531-1538.
- Amato M., Markus D., Huppi P. et al. //Ann. Hematol. 1991. 63: 210-213.
- Ichiyama T., Hayashi T., Furukawa S. //Neurology. 1991. 46(3): 837-838.
- Raes M., Alliet P., Gillis P. et al. // J. Pediatr. 1993. 123(3): 465-467.
- Ridge JP, Fuchs EJ, Matzinger P.//Science. 1996. 271: 1723-6.
- Schaffer C., Blanche-Ganter E. //Krankenhauspharmazie. 1998, 19. Lahrgang. No. 6. P. 280-286.
- Alejandria AA, Lansang MA, Dans LF et al.//Intravenous immunoglobulin for treating sepsis and septic shock. In the Cochrane Library issue. 1999. 3. Oxford.
- On the organization and conduct of preventive vaccinations against hepatitis B for newborns in obstetric hospitals in Moscow. M., 2001.
Note!
- The immunomodulatory effect of IVIG is closely related to the presence of the Fc function of the immunoglobulin molecule, as well as the content of Ig G subpopulations (primarily the Ig G2 subpopulation, the synthesis of which is reduced in children of the first year of life) and Ig M.
- Administration of immunoglobulin should be carried out as early as possible after diagnosis and in sufficient volume.
- To achieve maximum effectiveness of anti-infective therapy for severe infection and sepsis, it is most advisable to combine antibacterial therapy with intravenous immunoglobulin.
- The overall main advantage of using intravenous immunoglobulin preparations is the ability to quickly and easily achieve high serum concentrations of immunoglobulins.