Composition and release form
Tablets - 1 tablet:
- Active ingredients: levofloxacin hemihydrate - 512.46 mg (corresponding to the content of levofloxacin - 500 mg);
- Excipients: microcrystalline cellulose - 44.69 mg, crospovidone - 7.85 mg, sodium stearyl fumarate - 1.41 mg, croscarmellose sodium - 6.15 mg, colloidal silicon dioxide - 15.38 mg, maltodextrin - 24.6 mg, magnesium stearate - 2.46 mg;
- Shell composition: Opadry orange 20A230018 - 15 mg (hydroxypropyl methylcellulose 2910 [hypromellose 6cP] (E464) - 6.6 mg, titanium dioxide (E171) - 1.375 mg, talc - 3.150 mg, hyprolose [hydroxypropylcellulose, klucel EF] ( E463) - 3.851 mg, sunset yellow dye (E110) - 0.024 mg).
3/5/7/10 pcs. — contour packaging, cardboard packs.
Composition per tablet:
| Active ingredient, mg: | ||
| Levofloxacin hemihydrate (in terms of levofloxacin) | 256,23 250,00 | 512,46 500,00 |
| Excipients, mg: | ||
| Lactitol | 300,00 | 600,00 |
| Crospovidone | 32,50 | 65,00 |
| Povidone K-17 | 10,00 | 20,00 |
| Sodium stearyl fumarate | 9,75 | 19,50 |
| Talc | 6,50 | 13,00 |
| Microcrystalline cellulose to obtain a tablet weight | 650,00 | 1300,00 |
| Excipients of the shell, mg: | until you get a tablet weighing | |
| 670,00 | 1340,00 | |
| Hypromellose | 9,52 | 19,04 |
| Titanium dioxide | 5,22 | 10,44 |
| Macrogol-4000 | 3,744 | 7,488 |
| Talc | 1,10 | 2,20 |
| Povidone K-17 | 0,416 | 0,832 |
Description of the dosage form
Film-coated tablets, pinkish-orange, oval, biconvex; When cut, it is white to light yellow in color.
pharmachologic effect
Levofloxacin is a synthetic broad-spectrum antibacterial drug from the group of fluoroquinolones, containing levofloxacin, a levorotatory isomer of ofloxacin, as an active substance. Levofloxacin blocks DNA gyrase, disrupts supercoiling and cross-linking of DNA breaks, inhibits DNA synthesis, and causes profound morphological changes in the cytoplasm, cell wall and membranes.
Levofloxacin is active against most strains of microorganisms both in vitro and in vivo.
Aerobic gram-positive microorganisms: Corynebacterium diphtheriae, Enterococcus faecalis, Enterococcus spp, Listeria monocytogenes, Staphylococcus coagulase-negative methi-S(I), Staphylococcus aureus methi-S, Staphylococcus epidermidis methi-S, Staphylococcus spp(CNS), Streptococci group C and G, Streptococcus agalactiae, Streptococcus pneumoniae peni I/S/R, Streptococcus pyogenes, Viridans streptococci peni-S/R.
Aerobic gram-negative microorganisms: Acinetobacter baumannil, Acinetobacter spp, Actinobacillus actinomycetemcomitans, Citrobacter freundii, Eikenella corrodens, Enterobacter aerogenes, Enterobacter agglomerans, Enterobacter cloacae, Enterobacter spp, Escherichia coli, Gardnerella vaginalis, Haemophilus ducreyi, Haemophilus influenzae ampi-S/R, Haemophilus parainfluenzae, Helicobacter pylori, Klebsiella oxytoca, Klebsiella pneumoniae, Klebsiella spp, Moraxela catarrhalis (3+/p-, Morganella morganii, Neisseria gonorrhoeae non PPNG/PPNG, Neisseria meningitidis, Pasteurella conis, Pasteurella dagmatis, Pasteurella multocida, Pasteurella spp, Proteus mirabilis, Proteus vulgaris, Providencia rettgeri, Providencia stuartii, Providencia spp, Pseudomonas aeruginosa, Pseudomonas spp, Salmonella spp, Serratia marcescens, Serratia spp.
Anaerobic microorganisms: Bacteroides fragilis, Bifidobacterium spp, Clostridium perfringens, Fusobacterium spp, Peptostreptococcus, Propionibacterum spp, Veilonella spp.
Other microorganisms: Bartonella spp, Chlamydia pneumoniae, Chlamydia psittaci, Chlamydia trachomatis, Legionella pneumophila, Legionella spp, Mycobacterium spp, Mycobacterium leprae, Micobacterium tuberculosis, Mycoplasma hominis, Mycoplasma pneumoniae, Ricketsia spp, Ureaplasma urealyticum.
Pharmacokinetics
Levofloxacin is rapidly and almost completely absorbed after oral administration. Food intake has little effect on the speed and completeness of absorption. The bioavailability of 500 mg levofloxacin after oral administration is almost 100%. After taking a single dose of 500 mg of levofloxacin, Cmax is 5.2-6.9 mcg/ml, the time to reach Cmax is 1.3 hours, T1/2 is 6-8 hours.
Bonding with plasma proteins is 30-40%. Penetrates well into organs and tissues: lungs, bronchial mucosa, sputum, genitourinary organs, bone tissue, cerebrospinal fluid, prostate gland, polymorphonuclear leukocytes, alveolar macrophages.
In the liver, a small portion is oxidized and/or deacetylated. It is excreted from the body primarily by the kidneys by glomerular filtration and tubular secretion. After oral administration, approximately 87% of the dose taken is excreted unchanged in the urine within 48 hours, less than 4% in the feces within 72 hours.
Indications for use of Levofloxacin
Infectious and inflammatory diseases caused by sensitive microorganisms:
- acute sinusitis;
- exacerbation of chronic bronchitis;
- community-acquired pneumonia;
- complicated urinary tract infections (including pyelonephritis);
- uncomplicated urinary tract infections;
- prostatitis;
- infections of the skin and soft tissues;
- septicemia/bacteremia associated with the above indications;
- intra-abdominal infection.
Contraindications to the use of Levofloxacin
- hypersensitivity to levofloxacin or other quinolones;
- renal failure (with creatinine clearance less than 20 ml/min. - due to the impossibility of dosing this dosage form);
- epilepsy;
- tendon lesions due to previous treatment with quinolones;
- childhood and adolescence (up to 18 years);
- pregnancy and lactation period.
It should be used with caution in the elderly due to the high likelihood of a concomitant decrease in renal function, as well as in cases of glucose-6-phosphate dehydrogenase deficiency.
Use of Levofloxacin during pregnancy and children
The incidence of levofloxacin-related adverse reactions in Phase 3 clinical trials conducted in North America was 6.3%. Therapy was discontinued due to drug-related side effects in 3.9% of patients.
In clinical studies, the following side effects were considered likely to be related to levofloxacin: nausea (1.3%), diarrhea (1%), vaginitis (0.7%), insomnia (0.5%), abdominal pain (0. 4%), flatulence (0.4%), itching (0.4%), dizziness (0.3%), dyspepsia (0.3%), rash (0.3%), genital candidiasis (0. 2%), taste disturbance (0.2%), vomiting (0.2%), constipation (0.1%), fungal infection (0.1%), itching in the genital area (0.1%), headache pain (0.1%), thrush (0.1%), nervousness (0.1%), erythematous rash (0.1%), urticaria (0.1%).
In clinical studies, the following side effects were observed without taking into account the relationship with the drug.
From the nervous system and sensory organs: headache (6.4%), insomnia (4.6%), dizziness (2.7%), fatigue (1.2%), impaired taste sensitivity (1%); <1%: asthenia, incoordination, coma, convulsions, speech impairment, stupor, tremor, vertigo, confusion, aggression, agitation, anxiety, anorexia, delirium, depression, emotional lability, hallucinations, difficulty concentrating, mania, nervousness, paranoia, impaired thinking, unusual dreams, sleep disturbance, somnolence, diplopia, cerebrovascular disorders, tinnitus, hearing and vision impairment, conjunctivitis, parosmia.
From the cardiovascular system and blood <1%: hypertension, hypotension (including orthostatic), heart failure, circulatory failure, arrhythmia, bradycardia, tachycardia, blockade, cardiac arrest, supraventricular tachycardia, ventricular and atrial fibrillation, palpitation , angina pectoris, coronary thrombosis, myocardial infarction, thromboembolism, phlebitis, platelet pathology, epistaxis, purpura, thrombocytopenia, leukocytosis, leukopenia, lymphopenia, granulocytopenia, lymphadenopathy.
From the respiratory system: sinusitis (1.3%), rhinitis (1%); <1%: asthma, acute respiratory distress syndrome, cough, hemoptysis, dyspnea, hypoxia, pleural effusion, respiratory failure.
From the gastrointestinal tract: nausea (7.2%), diarrhea (5.6%), constipation (3.2%), abdominal pain (2.5%), dyspepsia (2.4%), vomiting (2 .3%), flatulence (1.5%); <1%: dry mouth, dysphagia, swelling of the tongue, gastroenteritis, gastrointestinal bleeding, pseudomembranous colitis, hepatic coma, increased LDH, jaundice, impaired liver function, cholelithiasis.
From the genitourinary system: vaginitis (1.8%); <1%: itching in the genital area, impaired ejaculation, impotence, increased serum creatinine, decreased renal function, acute renal failure, hematuria.
From the musculoskeletal system <1%: arthralgia, arthritis, arthrosis, muscle weakness, myalgia, osteomyelitis, synovitis, tendonitis, rhabdomyolysis, hyperkinesis, involuntary muscle contractions, increased muscle tone, paresthesia, paralysis.
From the skin: itching (1.3%), rash (1.2%); <1%: erythema nodosum, skin peeling, skin ulceration, urticaria, increased sweating.
Other: local reaction (3.5%), pain (1.7%) and inflammation (1.1%) at the injection site; pain (1.4%), chest pain (1.2%) and back pain (1.1%); <1%: hyperkalemia, hypokalemia, dehydration, hypoglycemia, hyperglycemia, worsening diabetes mellitus, weight loss, carcinoma, fever, facial edema, withdrawal syndrome.
The following adverse effects have been reported in post-marketing studies: hypersensitivity pneumonitis, anaphylactic shock, anaphylactoid reaction, dysphonia, abnormal EEG, encephalopathy, eosinophilia, erythema multiforme, hemolytic anemia, multiple organ failure, increased international normalized ratio (INR), Stevens-Johnson syndrome, rupture tendons, ventricular fibrillation, vasodilation.
Below are data from 29 pooled Phase 3 clinical trials (n=7537). The average age of patients is 50 years (approximately 74% of patients are younger than 65 years), 50% are male, 71% are Caucasian, and 19% are black. Patients received levofloxacin for the treatment of various infections at a dose of 750 mg once daily, 250 mg once daily, or 500 mg twice daily. The duration of therapy was usually 3–14 days (average 10 days).
The overall incidence, type, and distribution of adverse reactions were similar in patients receiving levofloxacin 750 mg once daily compared with patients receiving 250 mg once daily or 500 mg twice daily. Therapy was discontinued due to drug-related adverse events in 4.3% of patients overall, 3.8% of patients taking the 250 and 500 mg doses, and 5.4% of patients taking the 750 mg dose. The most common side effects leading to discontinuation of the drug at doses of 250 and 500 mg were gastrointestinal complaints (1.4%), nausea (0.6%), vomiting (0.4%), dizziness (0.3%) , headache (0.2%). The most common side effects leading to discontinuation of the drug at a dose of 750 mg were gastrointestinal disturbances (1.2%), nausea (0.6%), vomiting (0.5%), dizziness (0.3%), headache pain (0.3%).
The following are side effects noted in clinical trials and observed with an incidence of more than 0.1%.
From the nervous system and sensory organs: headache (6%), dizziness (3%), insomnia (4%); 0.1–1%: anxiety, agitation, confusion, depression, hallucinations, nightmares, sleep disturbances, anorexia, unusual dreams, tremors, convulsions, paresthesia, vertigo, hypertension, hyperkinesis, incoordination, drowsiness, fainting.
From the cardiovascular system and blood: 0.1–1%: anemia, arrhythmia, palpitations, cardiac arrest, supraventricular tachycardia, phlebitis, epistaxis, thrombocytopenia, granulocytopenia.
From the respiratory system: shortness of breath (1%).
From the gastrointestinal tract: nausea (7%), diarrhea (5%), constipation (3%), abdominal pain (2%), dyspepsia (2%), vomiting (2%); 0.1–1%: gastritis, stomatitis, pancreatitis, esophagitis, gastroenteritis, glossitis, pseudomembranous colitis, liver dysfunction, increased levels of liver enzymes, increased alkaline phosphatase.
From the genitourinary system: vaginitis (1%); 0.1–1%: impaired renal function, acute renal failure, genital candidiasis.
From the musculoskeletal system: 0.1–1%: arthralgia, tendinitis, myalgia, skeletal muscle pain.
From the skin: rash (2%), itching (1%); 0.1–1%: allergic reactions, edema (1%), urticaria.
Other: candidiasis (1%), reaction at the IV injection site (1%), chest pain (1%); 0.1–1%: hypoglycemia/hyperglycemia, hyperkalemia.
The following side effects have been reported in post-marketing studies.
From the nervous system and sensory organs: isolated reports of encephalopathy, EEG abnormalities, peripheral neuropathy, psychosis, paranoia, isolated reports of suicide attempts and suicidal thoughts, visual impairment (including diplopia, decreased visual acuity, blurred vision, scotoma ), hearing loss, tinnitus, parosmia, anosmia, loss of taste, taste perversion, dysphonia.
From the cardiovascular system and blood: isolated reports of torsade de pointes, prolongation of the QT interval, tachycardia, vasodilation, increased INR, prolongation of prothrombin time, pancytopenia, aplastic anemia, leukopenia, hemolytic anemia, eosinophilia.
From the gastrointestinal tract: liver failure (including fatal cases), hepatitis, jaundice.
From the musculoskeletal system: tendon rupture, muscle damage, including rupture, rhabdomyolysis.
From the skin: bullous rash, Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, photosensitivity/phototoxicity reactions.
Allergic reactions: hypersensitivity reactions (sometimes fatal), incl. anaphylactic/anaphylactoid reactions, anaphylactic shock, angioedema, serum sickness; isolated reports of hypersensitivity pneumonitis.
Other: vasculitis, increased activity of muscle enzymes, hyperthermia, multiorgan failure, interstitial nephritis.
When using levofloxacin in the form of 0.5% eye drops, the most frequently observed effects were: 1-3% - transient decreased vision, transient burning, pain or discomfort in the eye, sensation of a foreign body in the eye, fever, headache, pharyngitis, photophobia; <1% - allergic reactions, swelling of the eyelids, dry eyes, itching in the eye.
Fluoroquinolones, first approved for medical use in the 1980s, are among the most widely used antibacterial drugs. The combination of basic pharmacological properties (a wide spectrum of antimicrobial activity, an original mechanism of action, favorable pharmacokinetic properties and good tolerability) serves as the basis for their use in a wide range of community-acquired and nosocomial infections.
The introduction of various halogens and substituent groups into different positions of the quinolone core made it possible to synthesize a variety of drugs from the fluoroquinolone group of four generations with different benefit-risk ratios. The development of some representatives of this class was stopped at the pre-marketing stage, others were withdrawn in the post-marketing period due to safety problems, for example, tovafloxacin (hepatotoxicity) and grepafloxacin (development of life-threatening cardiac arrhythmias). A number of fluoroquinolones, in particular lomefloxacin and sparfloxacin, which have a higher risk of photo- and cardiotoxicity, have been displaced from the market in many countries by other drugs with a better benefit-risk ratio. Thus, only five fluoroquinolones remain on the US pharmaceutical market, and the volume of their prescriptions in recent years shows that some of them are also losing their clinical significance (Fig. 1). The only fluoroquinolone whose prescriptions have remained stable over the years is ciprofloxacin, and the only drug whose prescriptions continue to increase is levofloxacin [1]. Of the approximately 33 million fluoroquinolone prescriptions written in the United States in 2014, these 2 drugs accounted for approximately 22 million (ciprofloxacin - approximately 20 million, levofloxacin - 11.3 million), while the number of prescriptions for gemifloxacin was only 7006 , ofloxacin – 9500, and moxifloxacin – 609 thousand. A similar situation is observed in European countries, where prescriptions of ciprofloxacin and levofloxacin exceed 50% (median – 73%) of the prescriptions of all fluoroquinolones [2].
The basis for the increase in consumption of levofloxacin is its more favorable benefit-risk ratio compared to many other fluoroquinolones, as evidenced by the results of not only clinical, but also widely ongoing pharmacoepidemiological studies.
Pharmacological properties of levofloxacin
Levofloxacin has a wide spectrum of antibacterial action, including a large number of gram-positive and gram-negative aerobic microorganisms, incl. intracellular bacteria (Mycoplasma pneumonia, Chlamydia pneumoniae and Legionella pneumophila), Mycobacterium tuberculosis and Pseudomonas aeruginosa. It belongs to the so-called. respiratory fluoroquinolones, which are highly effective against the main pathogens of upper and lower respiratory tract infections, incl. Streptococcus pneumoniae, resistant to penicillins and macrolides. Levofloxacin is active against pathogens of the Enterobacteriacae family and, unlike most other drugs in its group, exhibits antipseudomonas activity, which serves as the basis for its prescription for nosocomial infections, incl. caused by multi-resistant microorganisms.
Levofloxacin is characterized by excellent pharmacokinetic properties, absolute (100%) bioavailability when taken orally, and a fairly long half-life (6–8 hours), providing high concentrations of the drug in the blood exceeding the MIC90 for many microorganisms, including the main causative agents of respiratory tract infections and pathogens of the Enterobacteriacae family. , for more than 24 hours, a large volume of distribution (90–110 l) and good penetration into the ENT organs, lungs, bronchial mucosa, sputum, bronchoalveolar fluid, prostate gland and prostatic fluid, gynecological organs, liver, biliary tract, skin , bones and joints. Excreted primarily by the kidneys, it creates high concentrations in the urine. The drug penetrates well into phagocytes and accumulates intracellularly [2].
The combination of a wide spectrum of action and pharmacokinetic properties ensures the effectiveness of levofloxacin for infections of various localizations, including nosocomial and community-acquired pneumonia, infections of the upper respiratory tract, skin and soft tissues, and urinary tract; chronic prostatitis and drug-resistant tuberculosis.
Safety and tolerability of levofloxacin
In recent decades, increasing attention has been paid to the safety issues of fluoroquinolones, which have a significant impact on the choice of a particular drug for a particular patient. Thanks to the development of pharmacovigilance systems in different countries and the introduction of electronic patient databases, the ability to assess the safety and tolerability of drugs not only in clinical trials, but also in real clinical practice has significantly expanded. Below are data on the safety of levofloxacin in comparison with other antibacterial agents, primarily other fluoroquinolone drugs, as well as risk factors for the development of rare serious adverse reactions (ARs) of drugs in this group, which must be taken into account to improve patient safety.
In clinical studies involving approximately 7000 patients with skin, respiratory and urinary tract infections, levofloxacin was at least as tolerable as amoxicillin/clavulanate, imipenem, clarithromycin, azithromycin, ceftriaxone, cefuroxime axetil, cefdinir and cefaclor [3], and the frequency of its serious ADRs in real medical practice per 1 million prescriptions in the first 39 months of being on the US market (15 million prescriptions) was: tendon ruptures - 4, taste disturbances - 3, convulsions - 2, photosensitivity - 1, hepatitis - 1 , liver failure – 1, QT interval prolongation – 1, torsade de pointes – 1, empyema – 1.7 [4]. An analysis of the Italian Pharmacovigilance database also showed that the relative risk of developing adverse reactions with levofloxacin, ciprofloxacin and norfloxacin did not differ from that for other classes of oral antibiotics, while cinoxacin and moxifloxacin were associated with a higher risk [5].
In clinical trials of levofloxacin, the most common adverse event was diarrhea, occurring in 4–6%, which is significantly lower than with oral penicillins or cephalosporins [6]. Adverse reactions from the central nervous system (headache, dizziness and sleep disturbances), which are a class effect of fluoroquinolones, were observed less frequently during treatment with levofloxacin (1%) than during treatment with gatifloxacin and moxifloxacin (2–3%).
Serious adverse reactions from the central nervous system, in particular seizures when using any fluoroquinolones, including levofloxacin, mainly occur in patients with risk factors for their development: epilepsy, traumatic brain injury, hypoxia, metabolic disorders, and self-resolve after discontinuation of the drug [7]. A risk factor for the development of seizures is also the concomitant use of non-steroidal anti-inflammatory drugs.
Another potential risk common to all fluoroquinolones is phototoxicity. The highest potential for phototoxicity is possessed by drugs that have a halogen atom in the 8th position of the quinolone ring - fleroxacin, clinafloxacin, sparfloxacin and lomefloxacin [8]. Thus, when using the last two drugs it is 8–10% or more [7, 9], while when using levofloxacin it does not exceed 0.1% [8].
The incidence of skin rashes with levofloxacin is 0.2% compared to 2.0% with moxifloxacin, 2.8–4.8% with gemifloxacin, 5.1% with sparfloxacin [8, 10]. Serious allergic reactions with its use are extremely rare. Over the almost 20-year period of widespread use of levofloxacin in real medical practice, only 4 cases of toxic epidermal necrolysis have been published [10]. The frequency of anaphylactoid reactions during treatment with levofloxacin is comparable to that with the use of most drugs in this group - 1.8–23 per 1 million days of treatment [10], and, apparently, significantly lower than when using moxifloxacin [11, 12]. Thus, according to the German Pharmacovigilance authorities, among 166 spontaneous reports of anaphylaxis with fluoroquinolones, 54% were associated with the use of moxifloxacin [13]. The incidence of anaphylaxis per 1 million daily doses was 3.3 for moxifloxacin, while for levofloxacin it was 0.6, and for ciprofloxacin and ofloxacin it was 0.2 each. It should be noted that some experts associate the increase in the frequency of hypersensitivity reactions to fluoroquinolones in the last decade with the introduction of moxifloxacin into medical practice [11, 12].
Hepatotoxic reactions with the use of fluoroquinolones are predominantly idiosyncratic (independent of dose and unpredictable) in nature. An increase in AST and ALT levels is observed in 2–3% of patients receiving drugs in this group.
In clinical studies of levofloxacin, hepatobiliary lesions (impaired liver function, increased levels of liver enzymes and alkaline phosphatase) occurred in 0.1–1% of cases [10]. The incidence of hepatitis, liver necrosis and liver failure during treatment with levofloxacin, according to French Pharmacovigilance data, is <1 5=»» 14=»» fda=»» 2=»» 1=»» 10=»» 58=»» 10= »» 6=»» 6=»» 6=»» 0=»» 15=»» 9=»» 16=»» 2008=»» ema=»» 2010=»» 17=»» 18=»» p=»»>
Fluoroquinolones are among the antibacterial agents associated with the highest risk of musculoskeletal injury, possibly due to their ability to chelate with di- and trivalent cations and have a toxic effect on collagen. When used, damage to tendons, cartilage, bones and muscles is possible.
Diffuse muscle pain, often in combination with muscle weakness, usually occurs in the first week of treatment and in most cases resolves spontaneously within 1–4 weeks after discontinuation of fluoroquinolones. However, there are reports of persistence of muscle pain for 6 months or more [19]. The risk of severe muscle damage, including rhabdomyolysis, increases with concomitant use of statins [20, 21], as well as in patients with underlying myopathy and myasthenia gravis [22, 23]. When using fluoroquinolones in patients with myasthenia, severe exacerbations have been described, incl. with fatal outcomes [23]. Comparative data on the incidence of muscle lesions with the use of different fluoroquinolone preparations could not be found in the available literature.
Concerns about the chondrotoxic effects of fluoroquinolones are based on experimental studies that have shown irreversible cartilage damage in fast-growing postnatal animals. Data on the development of arthropathy in humans are contradictory, incl. due to the lack of a unified definition of the term “arthropathy” and its use to describe various adverse reactions from the musculoskeletal system [6]. The results of numerous magnetic resonance imaging studies are inconsistent and difficult to interpret due to possible confounding factors, and there are no targeted randomized clinical trials examining chondrotoxicity in children.
Data from epidemiological studies are also contradictory. An increased risk of joint damage with fluoroquinolones was not observed in three of the four retrospective studies reviewed by Forsythe and Ernst (2007), although one of these studies found a correlation between pefloxacin use and the development of arthropathies [24]. A five-year follow-up of children (n=6000) taking antibiotics showed that the incidence of tendon or joint injuries with levofloxacin, ofloxacin or ciprofloxacin was <1% and comparable to that with azithromycin [25]. A systematic review assessing the efficacy and safety of ciprofloxacin in neonates found no evidence of osteoarticular toxicity [26]. Levofloxacin, according to the results of an analysis of clinical studies involving children older than 6 months with community-acquired pneumonia and otitis media, more often (1.6%) caused musculoskeletal lesions than β-lactam antibiotics and macrolides (0.7%), but the resulting The results may have been influenced by the inclusion of children with underlying joint diseases in these studies [27].
Thus, the available data do not allow us to unequivocally confirm the presence of an increased risk of arthropathy in children when treated with the most widely used fluoroquinolone drugs. However, childhood and pregnancy are contraindications to the use of fluoroquinolones.
In adult patients, fluoroquinolone-associated arthralgia is estimated to occur in 1% of cases, but its exact prevalence is unknown. The relationship between arthralgia and the presence of initial structural changes in the patient also remains unclear. There have been no studies assessing the risk of developing arthropathy itself in adult patients.
Tendinopathy (tendonitis and rupture of the tendon, mainly Achilles) has been described during treatment with all fluoroquinolone preparations for systemic use. According to systematic reviews, tendinopathy is slightly more common with ofloxacin, pefloxacin and ciprofloxacin than with other drugs in this group [28, 29]. The risk of their development when using ofloxacin and levofloxacin depends on the dose and duration of use [30].
In epidemiological studies, the incidence of tendinopathies with the use of fluoroquinolones was 1:2000 [6], according to the FDA - 1.3-5.6 per 10 thousand patient-years [31]. However, in patients with risk factors (see table), the likelihood of their development increases significantly. For example, in people who have undergone a kidney transplant, it can reach 12.2–15.6% [32], and in people over 80 years of age it can increase by 20.4 times [33]. If adequate therapeutic measures are not taken at the first symptoms, the lesion often progresses to tendon rupture. Approximately half of patients with tendon rupture had a recent history of glucocorticosteroid use [28].
Experiments on animals revealed the inhibitory effect of fluoroquinolones on the healing of bone fractures [34]. The clinical significance of this finding remains unclear, but until it is clarified, the authors recommended against the use of fluoroquinolones in the perioperative period in patients undergoing arthroplasty.
Fluoroquinolones, along with macrolides and azoles, are among the antimicrobial drugs whose use is most often associated with prolongation of the QT interval on the ECG [36]. Significant prolongation of the QT interval contributes to the development of torsades de pointes, a potentially fatal polymorphic ventricular tachycardia.
Due to the increased risk of developing torsade de pointes, grepafloxacin and the original drug sparfloxacin were withdrawn from the global pharmaceutical market. The mechanism of QT interval prolongation is associated with blockade of hERG potassium channels ((human ether-a-go-go-related), primarily leading to inhibition of the fast component of the potassium current rectifier (IKr) from myocytes, accumulation of potassium ions in myocytes and slowing of ventricular repolarization .
Fluoroquinolone preparations differ in their ability to inhibit hERG channels and, as a consequence, in their ability to induce torsade de pointes [37]. According to the degree of inhibitory effect on hERG channels, fluoroquinolones can be ranked in the following order: sparfloxacin > grepafloxacin > moxifloxacin > gatifloxacin > levofloxacin > ciprofloxacin > ofloxacin, which coincides with the ranking of fluoroquinolones according to the frequency of recording QT prolongation on the ECG [10].
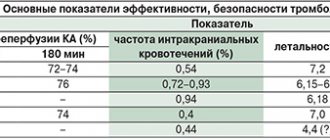
The risk of developing life-threatening arrhythmias with different fluoroquinolone drugs, according to the results of a population-based study conducted in Canada in 1990–2007. (1838 cases) [38], presented in Fig. 2.
The average QT prolongation caused by fluoroquinolones (3–6 ms) in patients with a normal QT interval (450–470 ms) is not of significant clinical significance. However, in individuals with a baseline QT interval >500 ms, fluoroquinolones should be avoided.
Risk factors for QT prolongation include:
- female;
- elderly age;
- hypokalemia, severe hypomagnesemia;
- bradycardia;
- recent cardioversion for atrial fibrillation, especially with QT prolonging drugs;
- chronic heart failure;
- left ventricular hypertrophy;
- ventricular arrhythmia;
- digoxin treatment;
- simultaneous use of 2 drugs that prolong QT;
- hereditary predisposition to QT prolongation [6, 40].
Since the degree of prolongation of the QT interval depends on the concentration of fluoroquinolones, the risk of arrhythmia increases under the influence of factors that increase the concentrations of these drugs in the blood or reduce their clearance, including. in case of impaired renal or liver function, against the background of drug interactions, with very rapid intravenous administration [6, 39].
Results from a recently published pilot study suggest that the risk of levofloxacin affecting the QT interval is dependent on circadian rhythms and may be reduced by adapting the timing of its administration to daily variations in cardiovascular parameters [40]. The maximum increase in the risk of QT interval prolongation was observed when the drug was administered at 2:00 p.m., the minimum at 6:00 a.m., however, these data require confirmation in further studies.
A number of studies have identified an association between the use of fluoroquinolones and disturbances in glucose homeostasis. The maximum risk of hypoglycemia is characteristic of gatifloxacin [41, 42]. In a comparative study, the risk of hypoglycemia with gatifloxacin was 2.81 times higher than that with levofloxacin [41]. In addition, gatifloxacin was associated with a high risk of hyperglycemia (odds ratio, 16.7), while no risk of this complication was identified with the use of other fluoroquinolones [42, 43]. The increased risk of dysglycemia, combined with the risk of hallucinations, drug-induced liver injury, and purpura, led to discontinuation of gatifloxacin in several countries [32].
The good tolerability of levofloxacin is also evidenced by the results of a pharmacoepidemiological study in the USA, which assessed the frequency of outpatient visits to medical care for ADRs of fluoroquinolones [44]. The estimated number of visits per year per 10 thousand population was minimal when using ciprofloxacin - 6.4 (95% confidence interval [CI] - 4.5-8.4), maximum when using moxifloxacin - 20.7 (95% CI – 11.9–29.5), during treatment with levofloxacin it was 8.9 (95% CI – 6.2–11.5).
Thus, levofloxacin has a wide spectrum of antibacterial activity against a variety of gram-positive and gram-negative microorganisms, including P. aeruginosa, excellent pharmacokinetic properties that allow it to achieve high concentrations in foci of infection of various locations and exhibit activity against intracellular pathogens, and good tolerability, at least not inferior to those of antibiotics from other widely used groups, and in many cases superior to it, which determines the favorable benefit-risk ratio of the drug for many community-acquired and nosocomial infections.
The risk of developing serious adverse reactions with levofloxacin is generally lower than with other fluoroquinolones and can be significantly reduced by rational use of the drug, which involves taking into account the risk factors for developing certain adverse reactions in a particular patient. The rational use of levofloxacin, as well as other fluoroquinolone drugs, will also reduce the rate of increase in antibiotic resistance to them, which is currently causing concern among the medical community. This is partly the focus of the FDA's recent warning to limit the use of fluoroquinolones for acute sinusitis, exacerbations of chronic bronchitis, and urinary tract infections for which alternative treatment options exist. In addition, the use of short 5-day courses of high dose levofloxacin (750 mg) is being considered as a measure to potentially reduce the risk of developing resistance [2, 45]. This treatment regimen allows you to create higher concentrations of the antibiotic in the blood, quickly eliminate the symptoms of infection and increase patient adherence to therapy. At the same time, the safety profile and frequency of side effects of levofloxacin when used in doses of 500 and 750 mg did not differ significantly in both clinical and pharmacoepidemiological studies.
In order to reduce the cost of treatment with levofloxacin, it is advisable to use its effective generics, in particular the drug Levolet R (Doctor Reddys Laboratories).
Side effects of Levofloxacin
Allergic reactions: sometimes – itching and redness of the skin; rarely - general hypersensitivity reactions (anaphylactic and anaphylactoid reactions) with symptoms such as urticaria, constriction of the bronchi and possibly severe suffocation; very rarely - swelling of the skin and mucous membranes (for example, in the face and throat), sudden drop in blood pressure and shock, increased sensitivity to solar and ultraviolet radiation, allergic pneumonitis, vasculitis; in some cases - severe skin rashes with blistering, for example, Stevens-Johnson syndrome, toxic epidermal necrolysis (Lyell's syndrome) and exudative erythema multiforme. General hypersensitivity reactions may sometimes be preceded by milder skin reactions. The above reactions can develop after the first dose, a few minutes or hours after administration of the drug.
From the digestive system: often - nausea, diarrhea, increased activity of liver enzymes (for example, alanine aminotransferase and aspartate aminotransferase); sometimes - loss of appetite, vomiting, abdominal pain, digestive disorders; rarely - diarrhea mixed with blood, which in very rare cases can be a sign of intestinal inflammation and even pseudomembranous colitis.
On the metabolic side: very rarely - a decrease in the concentration of glucose in the blood, which is of particular importance for patients with diabetes (possible signs of hypoglycemia: increased appetite, nervousness, perspiration, trembling). Experience with the use of other quinolones suggests that they can cause exacerbation of porphyria in patients already suffering from this disease. A similar effect cannot be excluded when using the drug levofloxacin.
From the nervous system: sometimes - headache, dizziness and/or stupor, drowsiness, sleep disturbances; rarely - anxiety, paresthesia in the hands, trembling, psychotic reactions such as hallucinations and depression, agitation, convulsions and confusion; very rarely - impaired vision and hearing, impaired taste and smell, decreased tactile sensitivity.
From the cardiovascular system: rarely - increased heartbeat, decreased blood pressure; very rarely - vascular (shock-like) collapse; in some cases - prolongation of the QT interval.
From the musculoskeletal system: rarely - tendon damage (including tendinitis), joint and muscle pain; very rarely - tendon rupture (for example, Achilles tendon); this side effect can be observed within 48 hours after the start of treatment and can be bilateral in nature, muscle weakness, which is of particular importance for patients with bulbar syndrome; in some cases - muscle damage (rhabdomyolysis).
From the urinary system: rarely - increased levels of bilirubin and creatinine in the blood serum; very rarely - deterioration of kidney function up to acute renal failure, interstitial nephritis.
From the hematopoietic organs: sometimes - an increase in the number of eosinophils, a decrease in the number of leukocytes; rarely - neutropenia, thrombocytopenia, which may be accompanied by increased bleeding; very rarely - agranulocytosis and the development of severe infections (persistent or recurrent increase in body temperature, deterioration in health); in some cases - hemolytic anemia; pancytopenia.
Other: sometimes - general weakness; very rarely - fever.
Any antibiotic therapy can cause changes in the microflora that is normally present in humans. For this reason, increased proliferation of bacteria and fungi resistant to the antibiotic used may occur, which in rare cases may require additional treatment.
Levofloxacin
Hospital-acquired infections caused by Pseudomonas aeruginosa may require combination treatment.
The prevalence of acquired resistance in cultured strains of microorganisms may vary by geographic region and over time. In this regard, information on drug resistance in a specific country is required.
For the treatment of severe infections or if treatment is ineffective, a microbiological diagnosis must be established with the isolation of the pathogen and determination of its sensitivity to levofloxacin.
Disability and potential irreversible serious adverse reactions associated with fluoroquinolones
The use of fluoroquinolones, including levofloxacin, has been associated with disability and the development of irreversible serious adverse reactions from various body systems that can develop simultaneously in the same patient.
Adverse reactions caused by fluoroquinolones include tendonitis, tendon rupture, arthralgia, myalgia, peripheral neuropathy, and nervous system side effects (hallucinations, anxiety, depression, insomnia, headaches, and confusion).
These reactions may develop from several hours to several weeks after starting levofloxacin therapy. The development of these adverse reactions was observed in patients of any age or without the presence of previous risk factors. If the first signs or symptoms of any serious adverse reactions occur, use of levofloxacin should be discontinued immediately. Fluoroquinolones, including levofloxacin, should be avoided in patients who have experienced any of these serious adverse reactions.
Methicillin-resistant streptococcus aureus
There is a high likelihood that methicillin-resistant Staphylococcus aureus will be resistant to fluoroquinolones, including levofloxacin. Therefore, levofloxacin is not recommended for the treatment of known or suspected infections caused by methicillin-resistant Staphylococcus aureus if laboratory tests have not confirmed the sensitivity of this microorganism to levofloxacin.
Patients predisposed to developing seizures
Like other quinolones, levofloxacin should be used with great caution in patients with a predisposition to seizures.
Such patients include patients with previous lesions of the central nervous system, such as stroke, severe traumatic brain injury; patients simultaneously receiving drugs that lower the seizure threshold of the brain, such as fenbufen and other similar non-steroidal anti-inflammatory drugs or other drugs that lower the seizure threshold, such as theophylline (see section "Interaction with other drugs").
Pseudomemdranosus colitis
Diarrhea that develops during or after treatment with levofloxacin, especially severe, persistent and/or bloody, may be a symptom of pseudomembranous colitis caused by Clostridium difficile. If pseudomembranous colitis is suspected, treatment with levofloxacin should be stopped immediately and specific antibiotic therapy (vancomycin, teicoplanin or oral metronidazole) should be started immediately. Drugs that inhibit intestinal motility are contraindicated.
Tendinitis and tendon rupture
Rarely observed, tendonitis with quinolones, including levofloxacin, can lead to rupture of tendons, including the Achilles tendon. This side effect can develop within 48 hours after starting treatment and can be bilateral. Elderly patients are more prone to developing tendonitis. The risk of tendon rupture may be increased when taking corticosteroids concomitantly. If tendinitis is suspected, treatment with Levofloxacin should be stopped immediately and appropriate treatment of the affected tendon should be initiated, for example by providing sufficient immobilization (see sections “Contraindications” and “Side effects”).
In addition, post-transplant patients have an increased risk of developing tendonitis, so it is recommended to be careful when prescribing fluoroquinolones to this category of patients.
In patients with impaired renal function, the daily dose should be adjusted based on creatinine clearance.
Application for airborne anthrax infection
The use of levofloxacin in humans for this indication is based on susceptibility data from Bacillus anthracis obtained from in vitro and experimental animal studies, as well as limited data from the use of levofloxacin in humans. Treating physicians should refer to national and/or international documents that reflect the collectively developed point of view on the treatment of anthrax.
Hypersensitivity reactions
Levofloxacin can cause serious, potentially fatal, hypersensitivity reactions (angioedema, anaphylactic shock), further with initial doses (see section "Side effects"). Patients should immediately stop taking the drug and consult a doctor.
Severe bullous reactions
Cases of severe bullous skin reactions such as Stevens-Johnson syndrome or toxic epidermal necrolysis have been observed while taking levofloxacin (see section "Side effects"). In case of development of any reactions from the skin or mucous membranes, the patient should immediately consult a doctor and not continue treatment until his consultation.
Disorders of the liver and biliary tract
Cases of hepatic necrosis, including the development of fatal liver failure, have been reported with the use of levofloxacin, mainly in patients with severe underlying diseases, such as sepsis (see section "Side effects").
Patients should be warned to stop treatment and seek immediate medical attention if signs and symptoms of liver damage occur, such as anorexia, jaundice, dark urine, itching and abdominal pain.
Patients with kidney failure
Since levofloxacin is excreted mainly through the kidneys, patients with impaired renal function require mandatory monitoring of renal function, as well as adjustment of the dosage regimen (see section "Dosage and Administration"). When treating elderly patients, it should be borne in mind that patients in this group often have impaired renal function (see section “Dosage and Administration”).
Preventing the development of photosensitivity reactions
Although photosensitivity develops very rarely with the use of levofloxacin, to prevent its development, patients are not recommended to be unnecessarily exposed to strong solar or artificial ultraviolet irradiation (for example, visiting a solarium) during treatment and for 48 hours after the end of treatment with levofloxacin.
Superinfection
As with the use of other antibiotics, the use of levofloxacin, especially for a long time, can lead to increased proliferation of microorganisms (bacteria and fungi) that are insensitive to it, which can cause changes in the microflora that is normally present in humans. As a result, superinfection may develop. Therefore, during treatment, it is imperative to re-evaluate the patient’s condition, and, if superinfection develops during treatment, appropriate measures should be taken.
QT prolongation
Very rare cases of QT prolongation have been reported in patients taking fluoroquinolones, including levofloxacin.
When using fluoroquinolones, including levofloxacin, caution should be exercised in patients with known risk factors for prolongation of the QT interval: in patients with uncorrected electrolyte disturbances (with hypokalemia, hypomagnesemia); with congenital long QT syndrome; with heart disease (heart failure, myocardial infarction, bradycardia); while taking medications that can prolong the QT interval, such as class IA and III antiarrhythmic drugs, tricyclic antidepressants, macrolides, antipsychotics.
Elderly and female patients may be more sensitive to drugs that prolong the QT interval. Therefore, fluoroquinolones, including levofloxacin, should be used with caution (see sections “With caution”, “Dosage and administration”, “Side effects” and “Overdose”, “Interaction with other drugs”).
Patients with glucose-6-phosphate dehydrogenase deficiency
Patients with latent or manifest glucose-6-phosphate dehydrogenase deficiency are predisposed to hemolytic reactions when treated with quinolones, which should be taken into account when treated with levofloxacin.
Hypo- and hyperglycemia (dysglycemia)
As with the use of other quinolones, cases of hyperglycemia and hypoglycemia have been observed with the use of levofloxacin, usually in patients with diabetes mellitus receiving concomitant treatment with oral hypoglycemic drugs (for example, glibenclamide) or insulin preparations. Cases of hypoglycemic coma have been reported.
In patients with diabetes mellitus, monitoring of blood glucose concentrations is required (see section "Side effects").
Peripheral neuropathy
Sensory and sensorimotor peripheral neuropathy, which may have a rapid onset, has been reported in patients taking fluoroquinolones, including levofloxacin. If the patient develops symptoms of neuropathy, levofloxacin should be discontinued. This minimizes the possible risk of developing irreversible changes.
Exacerbation of pseudoparalytic myasthenia gravis (myasthenia gravis)
Fluoroquinolones, including levofloxacin, have neuromuscular blocking activity and may increase muscle weakness in patients with myasthenia gravis. Adverse reactions, including pulmonary failure requiring mechanical ventilation and death, have been reported with the use of fluoroquinolones in patients with myasthenia gravis.
The use of levofloxacin in a patient with an established diagnosis of pseudoparalytic myasthenia gravis is not recommended (see “Side Effects”).
Psychotic reactions
With the use of quinolones, including levofloxacin, the development of psychotic reactions has been reported, which in very rare cases progressed to the development of suicidal thoughts and behavior disorders with self-harm (sometimes after taking a single dose of levofloxacin (see section "Side effects")). reactions, treatment with levofloxacin should be discontinued and appropriate therapy should be prescribed.
The drug should be prescribed with caution to patients with psychosis or patients with a history of mental illness.
Visual impairment
If any visual impairment develops, immediate consultation with an ophthalmologist is necessary (see section “Side Effects”).
Effect on laboratory tests
In patients taking levofloxacin, the determination of opiates in urine may lead to false-positive results, which should be confirmed by more specific methods.
Levofloxacin may inhibit the growth of Mycobacterium tuberculosis and subsequently lead to false-negative results of the bacteriological diagnosis of tuberculosis.
Drug interactions
There are reports of a pronounced decrease in the threshold of convulsive readiness with the simultaneous use of quinolones and substances that can, in turn, reduce the cerebral threshold of convulsive readiness. This also applies equally to the simultaneous use of quinolones and theophylline.
The effect of Levofloxacin is significantly weakened when used simultaneously with sucralfate. The same thing happens with the simultaneous use of magnesium or aluminum containing antacids, as well as iron salts. Levofloxacin should be taken at least 2 hours before or 2 hours after taking these medications. No interaction was detected with calcium carbonate.
When using vitamin K antagonists simultaneously, monitoring of the blood coagulation system is necessary.
The elimination (renal clearance) of levofloxacin is slightly slowed down by the action of cimetidine and probenecid. It should be noted that this interaction has virtually no clinical significance. However, with the simultaneous use of drugs such as probenecid and cimetidine, which block a certain excretion pathway (tubular secretion), treatment with levofloxacin should be carried out with caution. This applies primarily to patients with limited renal function.
Levofloxacin slightly increases the half-life of cyclosporine.
Taking glucocorticosteroids increases the risk of tendon rupture.
Directions for use and doses
Inside. Once or twice a day. The tablets should be swallowed without chewing and washed down with a sufficient amount of liquid (0.5 to 1 glass).
The drug can be taken before meals or at any time between meals, since food intake does not affect the absorption of the drug (see section "Pharmacokinetics").
The drug should be taken at least 2 hours before or 2 hours after taking drugs containing magnesium and/or aluminum, iron, zinc, or sucralfate (see section “Interaction with other drugs”).
Considering that the bioavailability of levofloxacin when taking Levofloxacin Ecolevid® tablets is 99-100%, if the patient is transferred from intravenous infusion with other levofloxacin drugs, taking Levofloxacin Ecolevid® tablets should be continued at the same dose that was used for intravenous infusion of levofloxacin drugs (see section "Pharmacokinetics").
Skipping one or more doses of the drug
If you accidentally miss a dose of the drug, you should take the next dose as soon as possible and then continue to take Levofloxacin Ecolevid® according to the recommended dosage regimen.
Levofloxacin dosage
The drug is taken orally 1 or 2 times a day. Do not chew the tablets and take a sufficient amount of liquid (from 0.5 to 1 glass); you can take them before meals or between meals. Doses are determined by the nature and severity of the infection, as well as the sensitivity of the suspected pathogen.
For patients with normal or moderately reduced renal function (creatinine clearance > 50 ml/min.), the following dosage regimen is recommended: sinusitis: 500 mg 1 time per day - 10-14 days; exacerbation of chronic bronchitis: 250 mg or 500 mg 1 time per day - 7-10 days; community-acquired pneumonia: 500 mg 1-2 times a day - 7-14 days. uncomplicated urinary tract infections: 250 mg 1 time per day for 3 days; prostatitis: 500 mg - 1 time per day - 28 days; complicated urinary tract infections, including pyelonephritis: 250 mg 1 time per day - 7-10 days; infections of the skin and soft tissues: 250 mg 1 time a day or 500 mg 1-2 times a day - 7-14 days; septicemia/bacteremia: 250 mg or 500 mg 1-2 times a day for 10-14 days; intra-abdominal infection: 250 mg or 500 mg 1 time per day - 7-14 days (in combination with antibacterial drugs acting on anaerobic flora).
Patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis do not require additional doses.
Patients with impaired liver function do not require special dosing, since levofloxacin is metabolized in the liver only to an extremely small extent.
As with the use of other antibiotics, treatment with Levofloxacin is recommended to be continued for at least 48-78 hours after normalization of body temperature or after laboratory confirmed recovery.
Levofloxacin: stepwise therapy for community-acquired pneumonia in adults
VC.
Duganov Main Military Clinical Hospital named after.
N.N. Burdenko, Moscow T
The traditional approach to the management of patients with severe or prognostically unfavorable lower respiratory tract infections (primarily pneumonia) involved parenteral administration of antibiotics throughout the entire period of hospitalization. At the same time, the alternative possibility of prescribing antibiotics in oral dosage form, which have a wide spectrum of antimicrobial activity, attractive oral bioavailability and are just as effective as parenteral forms of antibacterial drugs, was ignored to a certain extent. However, with the advent of new oral antibiotics, characterized by an excellent pharmacokinetic profile and safety, and the improvement of our knowledge of pharmacodynamic predictors of the effectiveness of antibacterial therapy, it has become possible to more often resort to prescribing oral antibiotics even for serious infections, incl. and respiratory tract.
Of particular interest in this case is the concept of the so-called. step therapy*
, which involves a two-stage use of antibacterial drugs: a transition from parenteral to non-parenteral (usually oral) route of administration in the shortest possible time, taking into account the clinical condition of the patient and without compromising the final effectiveness of treatment.
The main idea of step therapy is obvious advantages for the patient, the doctor and the medical institution (reducing the duration of the hospital period and transferring to treatment at home, which is psychologically more comfortable; minimizing the risk of nosocomial infections; reducing costs associated with the lower cost of oral antibiotics, eliminating additional costs for the administration of a drug in parenteral form, etc.) while maintaining high quality of medical care - corresponds with modern recommendations for the management of patients with community-acquired pneumonia [3, 4, 5], rightly emphasizing that currently the provision of highly effective/high-quality medical care should be carried out in the most economical way.
Stepped antibacterial therapy for pneumonia was first carried out in 1985, when F. Shann et al. [6] successfully used sequential administration of chloramphenicol in parenteral and then oral dosage forms in children of Papua and New Guinea. However, in fairness it must be said that only two years later R. Quintiliani et al. [7] presented the scientific rationale for this new approach to the use of antibacterial drugs.
In implementing the concept of stepwise antibacterial therapy, it is necessary to take into account several factors, namely the “patient factor”, “pathogen factor” and “antibiotic factor” [1] - see Table 1.
It is obvious that stepwise antibacterial therapy is not just a mechanical replacement of a parenteral drug with an oral one.
First of all, taking into account clinical expediency, the appropriate timing of this replacement must be determined. In this case, the main conditions for a safe transition to oral therapy
should be the following: a) the serious condition of the patient with unstable hemodynamics, requiring placement in the ICU, excludes the relevance of switching to oral antibiotics; b) normal gastrointestinal absorption; c) transfer to an oral antibiotic should be carried out at a time when it is possible to convincingly demonstrate an adequate clinical and laboratory “response” to the initiated parenteral (usually intravenous) therapy [5]. Typically, the latter include achieving apyrexia, reducing cough and the severity of other respiratory symptoms, a significant decrease in the number of peripheral blood leukocytes, etc. Thus, in particular, one of the widely popularized criteria for switching to oral antibacterial therapy for respiratory infections - JA Ramirez, 1995 [8] - are: reduction of cough and other respiratory symptoms; normal body temperature when measured sequentially at 8-hour intervals; a tendency towards normalization of the number of peripheral blood leukocytes; no disturbances in gastrointestinal absorption.
In general, based on an analysis of available studies assessing the effectiveness and safety of stepwise therapy for lower respiratory tract infections (primarily community-acquired pneumonia), the following conditions can be identified for switching to oral antibiotic use:
• achieving clinical improvement with initially administered intravenous antibiotic therapy;
• the patient has no known risk factors for an unfavorable prognosis of community-acquired pneumonia: condition after splenectomy, chronic alcoholism, intellectual and mental status disorders, serious deviations in the results of physical/laboratory examination - tachypnea ≥ 30/min, systolic blood pressure < 90 mm Hg. Art., hyperpyrexia > 38.3 °C, arterial hypoxemia < 60 mm Hg, the need for artificial ventilation, metastatic “screenings” of infection (brain abscess, etc.), leukopenia (<4x109/l) or hyperleukocytosis (> 30x109/l), renal failure (residual urea nitrogen > 20 mg/dl), multilobar pneumonic infiltration, rapid progression of focal infiltrative changes in the lungs, destruction of lung tissue, signs of severe infectious process (metabolic acidosis, septic shock, respiratory distress syndrome of adults, etc.).
At the same time, the time for switching from the intravenous to the oral route of antibiotic administration varies, as a rule, from 48 to 72 hours. According to some publications, the next 48 hours seem to be the optimal time period for making a decision on switching to an oral antibiotic [10].
A simple, at first glance, scheme of stepwise antibacterial therapy for lower respiratory tract infections can sometimes be difficult to implement, since the patient may be in the field of view of doctors of various specialties (hence the widest possible popularization of modern recommendations for the management of patients with community-acquired pneumonia becomes extremely important). In this context, the possible features of doctor-patient cooperation should also be taken into account. And, finally, it is necessary to take into account the fact that in some patients there is a slow reverse clinical and radiological development of the disease, which means that before switching to oral therapy, an analysis of the possible causes of the protracted course of community-acquired pneumonia should be carried out.
To date, we have a very limited number of controlled clinical studies confirming the high effectiveness and safety of stepwise therapy for lower respiratory tract infections (Table 2). Nevertheless, the available data provide a strong argument in favor of, if possible, an early transition to oral antibiotics in cases where an adequate clinical and/or laboratory “response” is achieved against the background of initial intravenous therapy for community-acquired pneumonia [3,4,5].
When choosing a drug for oral administration as part of step-down therapy, preference should be given to those antibiotics that demonstrate an identical or close spectrum of antimicrobial activity to antibiotics administered parenterally. At the same time, most doctors feel more comfortable if the switch occurs to the oral form of the same antibiotic (on the contrary, the very fact that in some cases the corresponding antibiotic is not available in an oral dosage form may delay the timing of the planned “switch”). Of particular importance is the dosage regimen that corresponds to high or, conversely, low compliance. Antibiotics taken 1 or 2 times a day have additional benefits in this regard. The requirements for oral antibiotics should also include high bioavailability, an acceptable safety profile, and a minimal level of drug interactions.
All these requirements, especially in the context of the treatment of community-acquired pneumonia, are best met by levofloxacin
– from among the new or so-called respiratory fluoroquinolones.
First, like other new or “respiratory” fluoroquinolones (moxifloxacin, gatifloxacin, gemifloxacin), levofloxacin has a broad spectrum of activity against all potential pathogens of community-acquired pneumonia, including Streptococcus pneumoniae
(regardless of their sensitivity to penicillin and/or macrolides), atypical pathogens and gram-negative bacilli [22].
Secondly, levofloxacin is characterized by attractive pharmacokinetic parameters: almost absolute bioavailability when taken orally (i99%); achieving high and predictable concentrations in the bronchial mucosa, the fluid lining the bronchial epithelium, alveolar macrophages, polymorphonuclear leukocytes, exceeding the concentration in the blood serum [23, 24].
Thirdly, levofloxacin is available in dosage forms for intravenous administration and oral administration, prescribed once a day.
Fourth, levofloxacin has an acceptable safety profile, comparable to that of comparator drugs. Thus, in particular, levofloxacin is characterized by slight phototoxicity [25], the absence of serious adverse effects from the central nervous system [26], is not metabolized by enzymes of the cytochrome P450 system, and therefore does not interact with warfarin, theophylline, and is generally characterized by a minimal degree of medicinal properties. interactions [27]. When taking levofloxacin, prolongation of the corrected QT interval and clinically significant hepatotoxicity have not been established [28]. Since the registration of levofloxacin in the USA in 1997 (it has been used in Japan since 1993), extensive experience in the successful clinical use of this antibiotic has been accumulated throughout the world, covering more than 150 million patients [29]. This circumstance seems especially important, since the specific problems of individual fluoroquinolones (temafloxacin, trovafloxacin, grepafloxacin, clinafloxacin, lomefloxacin, sparfloxacin) could create the image of “toxic antibiotics” for the entire class.
To date, well-designed controlled studies have provided extensive evidence of similar or superior clinical and/or microbiological efficacy of levofloxacin compared to comparator antibiotics in stepwise therapy for community-acquired pneumonia. One study examined the clinical/microbiological efficacy and safety of levofloxacin administered intravenously (500 mg once daily) and/or orally (500 mg once daily) in patients with community-acquired pneumonia in comparison with ceftriaxone (1.0 –2.0 g 1–2 times/day) and (or) cefuroxime axetil (500 mg 2 times/day) [30]. In addition, based on the specific clinical situation, patients randomized to the Ceftriaxone ± cefuroxime axetil group could be prescribed erythromycin or doxycycline. This addition turned out to be very relevant, because Chlamydia pneumoniae–, Mycoplasma pneumoniae in a significant number of examined patients
– and
Legionella pneumophila
infections (in 101, 41 and 8 patients, respectively). In both groups, the duration of antibiotic therapy did not exceed 12 days. At the same time, 2% of patients received levofloxacin only in parenteral dosage form, 61% - orally, and 37% - as part of step-down therapy. In the comparison group, cephalosporins in parenteral, oral form and as part of step-down therapy were prescribed in 2%, 50% and 48% of cases, respectively.
A comparative analysis showed that the clinical and microbiological effectiveness of levofloxacin monotherapy (prescribed also as part of step-down therapy) was significantly higher than traditional treatment regimens for community-acquired pneumonia (ceftriaxone ± cefuroxime axetil ± erythromycin or doxycycline) with a comparable frequency of adverse events - 5. 8% and 8.5% respectively. Moreover, this superiority was in no way connected with the known advantage of fluoroquinolone over cephalosporins against “atypical” pathogens (Table 3).
Another study examined the comparative effectiveness of levofloxacin in stepwise therapy (500 mg 2 times/day*) and ceftriaxone (4.0 g 1 time/day) in patients with severe community-acquired pneumonia [31]. Evidence of the initially severe condition of the patients was a comparable “representation” of patients in both groups with an integral score on the APACHE II scale of 15 points (21%), as well as a mortality rate of 7%. In the group receiving levofloxacin, all patients received at least 4 doses of the drug intravenously, and the majority of patients (87%) eventually switched to taking the antibiotic orally.
The data obtained indicated comparable clinical and microbiological effectiveness of levofloxacin and ceftriaxone in the treatment of severe community-acquired pneumonia (Table 4), although there were significantly more cases of ceftriaxone discontinuation due to early clinical ineffectiveness (p = 0.05).
The role and place of levofloxacin in the stepwise therapy of community-acquired pneumonia in comparison with traditional therapy was studied as part of a large-scale Canadian study (
CAPITAL Study
), which included 1743 patients observed in 20 centers [32]. To resolve the issue of the place of treatment and method of drug administration, the well-known prognostic scale MJ Fine et al., 1997 [33] was used. Moreover, if, according to this scale, the patient’s final score did not exceed 90, then treatment was carried out at home with the prescription of levofloxacin (500 mg 1 time/day, orally) for 10 days. If the final score was 91 or more, then the patient was hospitalized, and initially levofloxacin (500 mg 1 time/day) was administered intravenously (the first dose within the next 4 hours from the moment the patient sought medical help). Subsequently, upon achieving a stable condition (ability to swallow food and liquid, negative blood cultures, body temperature Ј38.0°C, respiratory rate <24/min, heart rate <100/min), treatment continued with the administration of oral levofloxacin (500 mg 1 time/day). Since the cited study also examined the possibility of minimizing the hospital period of treatment against the background of step-by-step therapy with levofloxacin, it seemed practically important to identify unified criteria for discharging a patient from the hospital: a) criteria that allow “switching” to taking an antibiotic orally; b) the number of peripheral blood leukocytes <12×109/l; c) stable course of concomitant diseases; d) normal oxygenation (when breathing room air SaO2>90%) for patients with concomitant chronic obstructive pulmonary disease with pO2>60 mmHg. Art.
As the analysis of the results of the study showed, there were no significant differences in the frequency of re-hospitalization, mortality and quality of life (SF-36 rating scale) among patients with community-acquired pneumonia who received levofloxacin as part of stepwise therapy or standard treatment. At the same time, the introduction of stepwise therapy with levofloxacin led to a decrease in the length of patient stay in hospital by an average of 1.7 days, a decrease in bed days for this nosological form by 18% and a reduction in costs by $1,700 (per patient).
Finally, the results of another multicenter, open-label, randomized comparative study were recently published, which aimed to study the clinical and microbiological effectiveness of levofloxacin and ceftriaxone in combination with erythromycin in patients with community-acquired pneumonia at high risk of adverse outcome [34]. Evidence of the initial severe condition of the patients was the corresponding values of the final score on the APACHE II scale, which amounted to 15.9 ± 6.29 in the group of patients receiving levofloxacin, and 16.0 ± 6.65 in the comparison group.
In patients receiving levofloxacin (n=132), the drug was initially administered intravenously at a dose of 500 mg 1 time / day (i 24 hours), and then the antibiotic was continued in oral dosage form (500 mg 1 time / day) for 7-14 days. In the comparison group (n = 137), patients were administered intravenous or intramuscular ceftriaxone (1–2 g 1 time/day) and intravenous erythromycin (500 mg 4 times/day), followed by switching to oral amoxicillin/clavulanate (875 mg 2 times/day). day) in combination with clarithromycin (500 mg 2 times/day).
Integral clinical (cases of cure and clinical improvement) and microbiological effectiveness were comparable in both groups (Table 5).
Since most of the previously published studies analyzed cases of community-acquired pneumonia with a low risk of adverse outcome, it is obvious that this study provides unique information indicating that levofloxacin monotherapy is at least as effective as the traditional combination treatment of ceftriaxone + erythromycin in this category of patients with a high probability of death.
As mentioned above, such properties of levofloxacin as the possibility of administering the drug in parenteral and oral dosage forms, proven clinical effectiveness in the treatment of respiratory tract infections, almost absolute bioavailability, safety, absence of clinically significant drug interactions, good tolerability when taken orally, long dosing interval create the image of an “ideal” antibiotic for stepwise therapy of community-acquired pneumonia
. And in the studies conducted to date, including and patients with a severe and (or) prognostically unfavorable course of the disease, convincing evidence was obtained of superior or at least comparable clinical and microbiological effectiveness of levofloxacin monotherapy compared with traditional combination treatment (cephalosporins + macrolides). This circumstance, as well as an excellent safety profile, confirmed by many years of widespread clinical use, and the obvious economic advantages of monotherapy explain the presence of levofloxacin in modern treatment regimens for community-acquired pneumonia, especially in hospital settings (Fig. 1).
Rice.
1. Algorithm for the management of a patient with community-acquired pneumonia (according to JG Bartlett, 2001, as amended) 1 - levofloxacin, etc. (currently in Russia only levofloxacin is available in parenteral and oral dosage forms) 2 - cefotaxime or ceftriaxone 3 - erythromycin, clarithromycin or azithromycin (the dosage form for intravenous administration is at the stage of registration with the Ministry of Health of the Russian Federation) References:
1. Fine A., Grossman R., Ost D., Farber B., Cassiere H. Diagnosis and Management of Pneumonia and Other Respiratory Infections. 1st ed. Berlin:PCI; 1999.
2. Strachunsky L.S., Rozenson O.L. Stepped therapy: a new approach to the use of antibacterial drugs. Clinical Pharmacology and Therapy 1997; 6:15–24.
3. Bartlett JG, Breiman RF, Mandell LA, File TM Community–acquired pneumonia in adults: guidelines for management. Infectious Diseases Society of America. Clin Infect Dis 1998; 26:811–838.
4. Bartlett JG, Dowell SF, Mandell LA et al. Practice guidelines for the management of community–acquired pneumonia in adults. Infectious Diseases Society of America. Clin Infect Dis 2000; 31: 347–382.
5. Mandell LA, Marrie TJ, Grossman RF et al. Canadian guidelines for the initial management of community-acquired pneumonia: an evidence-based update by the Canadian Infectious Diseases Society and the Canadian Thoracic Society. Clin Infect Dis 2000; 31: 383–421.
6. Shann F., Barker J., Poore P. Chloramphenicol alone versus chloramphenicol plus penicillin for severe pneumonia in children. Lancet 1985; 2:684–685.
7. Quintiliani R, Cooper BW, Briceland LL et al. Economic impact of streamlining antibiotic administration. Am J Med 1987; 82(suppl 4A): 391–394.
8. Ramirez JA Switch therapy in community-acquired pneumonia. Diagn Microbiol Infect Dis 1995; 22: 219–223.
9. Nathwani D. Sequential Switch Therapy for Lower Respiratory Tract Infections. A European Perspective. Chest 1998; 113:211–218.
10. Weingarten SR, Reidinger MS, Varis G. et al. Identification of low-risk hospitalized patients with pneumonia: implications for early conversion to oral antimicrobial therapy. Chest 1994; 105:1109–1115.
11. Vogel F., Multicentre Trial Group. Efficacy and tolerance of cefotaxime followed by oral cefixime versus cefotaxime alone in patients with lower respiratory tract infections. Curr Ther res 1994; 55(suppl A): 42–48.
12. Khan FA, Basir R. Sequential intravenous–oral administration of ciprofloxacin vs ceftazidime in serious bacterial respiratory tract infections. Chest 1989; 96:528–537.
13. Paladino J., Sperry H., Backes J. et al. Clinical and economic evaluation of oral ciprofloxacin after an abbreviated course of intravenous antibiotics. Am J Med 1991; 91:462–470.
14. Niederman MS, Bass JB, Campbell AM et al. Guidelines for the initial management of adults with community–acquired pneumonia: diagnosis, assessment of severity, and initial antimicrobial therapy: American Thoracic Society, Medical Section of the American Lung Association. Am Rev Pespir Dis 1993; 148:1418–1426.
15. Brambilla C., Kastanakis S., Knight S. et al. Cefuroxime and cefuroxime axetil versus amoxicillin plus clavulanic acid in the treatment of lower respiratory tract infections. Eur L Clin Microb Infect Dis 1992; 11: 118–124.
16. Feist H. Sequential therapy with IV and oral ofloxacin in lower respiratory tract infections: a comparative study. Infection 1991; 19(suppl 7):380–383.
17. Khajalia R., Driicek M., Vetter N. A comparative study of loxacin and amoxycillin/clavulanate in hospitalized patients with lower respiratory tract infections. J Antimicrob Chemother 1990; 26 (suppl D): 83–91.
18. Ramirez JA, Srinath L, Ahkee S et al. Early switch from intravenous to oral cephalosporins in the treatment of hospitalized patients with community acquired pneumonia. Arch Intern Med 1995; 155:1273–1276.
19. Gentry LO, Rodriguez–Gomez G, Kohler RB et al. Parenteral followed by oral ofloxacin for nosocomial pneumonia and community–acquired pneumonia requiring hospitalization. Am Rev Respir Dis 1992; 145: 31–35.
20. Ramirez JA, Akhee S. Early switch from intravenous antimicrobials to oral clarithromycin in the treatment of hospitalized patients with community acquired pneumonia [abstract 12.04]. Abstracts of the Third International Conference on Macrolides, Azalides and Streptogramins, Lisbon, Portugal, 1996; 83.
21. Brande P., Vondra V., Vogel F. et al. Sequential therapy with cefuroxime followed by cefuroxime axetil in community–acquired pneumonia. Chest 1997; 112:406–415.
22. Davis R., Bryson HM Levofloxacin. A review of its antibacterial activity, pharmacokinetics and therapeutic efficacy. Drugs 1994; 47: 677–700.
23. Chien SC, Rogge MC, Gisclon LG et al. Pharmacokinetic profile of levofloxacin following once daily 500–milligram oral or intravenous doses. Antimicrob Agents Chemother 1997; 41:2256–2260.
24. Preston SL, Drusano GL, Berman AL et al. Levofloxacin population pharmacokinetics and a creation of demographic model for prediction of individual drug clearance in patients with serious community–acquired infection. Antimicrob Agents Chemother 1998; 42:1098–1104.
25. Lipsky BA, Baker CA Fluoroquinolone toxicity profiles: a review focusing on newer agents. Clin Infect Dis 1999; 28: 352–364.
26. Nau R., Kinzig M., Dreyhaupt T. et al. Kinetics of ofloxacin and its metabolites in cerebrospinal fluid after single intravenous infusion of 400 milligrams of ofloxacin. Antimicrob Agents Chemother 1994; 38: 1849–1853.
27. Fish DN, Chow AT The clinical pharmacokinetics of levofloxacin. Clin Pharmacokinet 1997; 32: 101–119.
28. Owens R., Ambrose P. Clinical use of the fluoroquinolones. Med Clin North Am 2000; 84:1447–1469.
29. Personal communication: https:// www.infectweb.com/
30. File TM, Sergeti J., Player R. et al. A multicenter, randomized study comparing the efficacy and safety of intravenous and/or oral levofloxacin versus ceftriaxone and/or cefuroxime axetil in the treatment of adults with community–acquired pneumonia. Antimicrob Agents Chemother 1997; 41: 1965–1972.
31. Norrby SR, Petermann W, Willcox PA et al. A comparative study of levofloxacin and ceftriaxone in the treatment of hospitalized patients with pneumonia. Scand J Infect Dis 1998; 30: 397–404.
32. Marrie TJ, Lau CY, Wheeler SL et al. Controlled trial of critical pathway for treatment of community-acquired pneumonia. JAMA 2000; 283:749–755.
33. Fine MJ, Auble TE, Yealy DM et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 1997; 336:243–250.
34. Kahn JB, Wiesinger A, Olson WH et al. Levofloxacin vs. ceftriaxone sodium and erythromycin in the treatment of patients with community–acquired pneumonia at high risk of mortality. Abstracts of 7th International Symposium on New Quinolones. Edinburgh, UK, 2001; 45.
35. Bartlett JG Respiratory Tract Infections. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2001.
Levofloxacin –
Tavanik (trade name)
(Aventis Pharma)
Overdose
Symptoms of an overdose of Levofloxacin appear at the level of the central nervous system (confusion, dizziness, disturbances of consciousness and seizures of the epileptic type). In addition, gastrointestinal disorders (for example, nausea) and erosive lesions of the mucous membranes, prolongation of the QT interval may occur.
Treatment should be symptomatic. Levofloxacin is not eliminated by dialysis (hemodialysis, peritoneal dialysis and continuous peritoneal dialysis). There is no specific antidote.