Metformin, 850 mg, film-coated tablets, 60 pcs.
Lactic acidosis
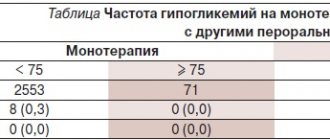
Lactic acidosis is a rare but serious (high mortality unless promptly treated) complication that may occur due to accumulation of metformin. Cases of lactic acidosis when taking metformin occurred mainly in patients with diabetes mellitus with severe renal failure.
Other associated risk factors should be taken into account, such as decompensated diabetes mellitus, ketosis, prolonged fasting, alcoholism, liver failure and any condition associated with severe hypoxia. This may help reduce the incidence of lactic acidosis.
The risk of developing lactic acidosis should be taken into account when nonspecific signs appear, such as muscle cramps accompanied by dyspeptic symptoms, abdominal pain and severe asthenia. Lactic acidosis is characterized by acidotic shortness of breath, abdominal pain and hypothermia followed by coma.
Diagnostic laboratory parameters are a decrease in blood pH (less than 7.25), lactate content in the blood plasma over 5 mmol/l, increased anion gap and lactate/pyruvate ratio.
If metabolic acidosis is suspected, stop taking the drug and consult a doctor immediately.
Surgical operations
The use of metformin should be discontinued 48 hours before elective surgery and can be continued no earlier than 48 hours after, provided that renal function was found to be normal during the examination.
Metformin should be replaced with another hypoglycemic drug (for example, insulin) 48 hours before an x-ray examination with intravenous contrast agents and continued for another 48 hours after this examination.
Kidney function
Since metformin is excreted by the kidneys, before starting treatment and regularly thereafter, it is necessary to determine CC: at least once a year in patients with normal renal function, and 2-4 times a year in elderly patients, as well as in patients with CC at the lower limit norms.
Particular caution should be exercised in case of possible impairment of renal function in elderly patients, with simultaneous use of antihypertensive drugs, diuretics or non-steroidal anti-inflammatory drugs.
Use in children and adolescents
The diagnosis of type 2 diabetes mellitus must be confirmed before starting treatment with metformin.
In clinical studies lasting 1 year, metformin was shown to have no effect on growth and puberty. However, due to the lack of long-term data, careful monitoring of the subsequent effects of metformin on these parameters in children, especially during puberty, is recommended. The most careful monitoring is necessary in children aged 10–12 years.
Other Precautions
Patients are advised to continue to follow a diet with even carbohydrate intake throughout the day. Overweight patients are recommended to continue to follow a hypocaloric diet (but not less than 1000 kcal/day).
It is recommended that routine laboratory tests be performed regularly to monitor diabetes mellitus.
It is not recommended to prescribe the drug if there is a risk of dehydration.
Metformin does not cause hypoglycemia when used alone, but caution is recommended when used in combination with insulin or other hypoglycemic agents (for example, sulfonylureas, repaglinide). When combined treatment requires careful monitoring of blood glucose concentrations.
Impact on the ability to drive vehicles and machinery
Monotherapy with the drug does not cause hypoglycemia and therefore does not affect the ability to drive vehicles and machines. Patients should be warned about the risk of developing hypoglycemia when using metformin in combination with other hypoglycemic drugs (sulfonylurea derivatives, insulin, repaglinide, etc.), which impair the ability to drive vehicles and engage in other potentially hazardous activities that require increased alertness and quick response. psychomotor reactions.
Metformin canon tablet p/o film 850mg 60 pcs
Contraindicated combinations Radiological studies using iodine-containing X-ray contrast agents can cause the development of lactic acidosis in patients with diabetes mellitus against the background of functional renal failure. The use of metformin should be discontinued 48 hours before and not resumed until 48 hours after an X-ray examination using radiocontrast agents.
Combinations not recommended
With the simultaneous use of metformin with alcohol and ethanol-containing drugs, during acute alcohol intoxication, during fasting or following a low-calorie diet, as well as in liver failure, the risk of developing lactic acidosis increases.
Combinations requiring special caution
With simultaneous use of metformin with danazol, a hyperglycemic effect may develop. If treatment with danazol is necessary and after stopping its use, a dose adjustment of metformin is required under the control of blood glucose concentrations.
Chlorpromazine in high doses (100 mg/day) reduces insulin release and increases blood glucose concentrations. When used simultaneously with antipsychotics and after stopping their use, a dose adjustment of metformin is required under the control of blood glucose concentrations.
Glucocorticosteroids (GCS), when administered parenterally and locally, reduce glucose tolerance and increase blood glucose concentrations, in some cases causing ketosis. If it is necessary to use such a combination and after stopping the use of corticosteroids, a dose adjustment of metformin is required under the control of blood glucose concentrations.
With the simultaneous use of loop diuretics and metformin, there is a risk of developing lactic acidosis due to the possible occurrence of functional renal failure.
The use of beta2-adrenergic agonists by injection reduces the hypoglycemic effect of metformin due to stimulation of beta2-adrenergic receptors. In this case, you should monitor the concentration of glucose in the blood and, if necessary, use insulin.
Angiotensin-converting enzyme inhibitors and other antihypertensive drugs may decrease blood glucose concentrations. If necessary, the dose of metformin should be adjusted.
With simultaneous use of metformin with sulfonylurea derivatives, insulin, acarbose and salicylates, the hypoglycemic effect may be enhanced.
Nifedipine increases the absorption and Cmax of metformin, which must be taken into account when using them simultaneously. Loop diuretics and nonsteroidal anti-inflammatory drugs (NSAIDs) increase the risk of decreased kidney function. In this case, caution must be exercised when using metformin.
Metformin
Lactic acidosis
Lactic acidosis is a rare but serious (high mortality unless promptly treated) complication that may occur due to accumulation of metformin. Cases of lactic acidosis when taking metformin occurred mainly in patients with diabetes mellitus with severe renal failure.
Other associated risk factors should be taken into account, such as decompensated diabetes mellitus, ketosis, prolonged fasting, alcoholism, liver failure, any condition associated with severe hypoxia and simultaneous use of drugs that can cause the development of lactic acidosis (see section “Interaction with other drugs”) "). This may help reduce the incidence of lactic acidosis.
The risk of developing lactic acidosis should be taken into account when nonspecific signs appear, such as muscle cramps accompanied by dyspeptic disorders, abdominal pain and severe asthenia. Lactic acidosis is characterized by acidotic shortness of breath, abdominal pain and hypothermia followed by coma. Diagnostic laboratory parameters are a decrease in blood pH (less than 7.35), lactate content in the blood plasma over 5 mmol/l, increased anion gap and lactate/pyruvate ratio. If metabolic acidosis is suspected, stop taking the drug and consult a doctor immediately.
Surgical operations
Metformin should be discontinued during surgery under general, spinal or epidural anesthesia. Metformin therapy can be continued no earlier than 48 hours after surgery or resumption of food intake, provided that renal function has been examined and found to be normal.
Kidney function
Since metformin is excreted by the kidneys, before starting treatment and regularly thereafter, it is necessary to determine the clearance of creatinan:
- At least once a year in patients with normal renal function;
- Every 3-6 months in patients with creatinine clearance 45-59 ml/min.
- Every 3 months in patients with creatinine clearance 30-44 ml/min.
In case of creatinine clearance less than 30 ml/min, the use of the drug is contraindicated.
Particular caution should be exercised in case of possible impaired renal function in elderly patients, with dehydration (chronic or severe diarrhea, repeated bouts of vomiting), and with the simultaneous use of antihypertensive drugs, diuretics or non-steroidal anti-inflammatory drugs.
Heart failure
Patients with heart failure have a higher risk of developing hypoxia and renal failure. Patients with chronic heart failure should have their cardiac renal function monitored regularly while taking metformin.
Taking metformin in heart failure with unstable hemodynamic parameters is contraindicated.
Children and teenagers
The diagnosis of type 2 diabetes mellitus must be confirmed before starting treatment with metformin.
In clinical studies lasting 1 year, metformin was shown to have no effect on growth and puberty. However, due to the lack of long-term data, careful monitoring of the subsequent effects of metformin on these parameters in children, especially during puberty, is recommended. The most careful monitoring is necessary for children aged 10-12 years.
The use of iodine-containing radiocontrast agents
Intravascular administration of iodinated radiocontrast agents can lead to the development of renal failure and accumulation of metformin, which increases the risk of developing lactic acidosis. Metformin should be discontinued, depending on renal function, 48 hours before or during an X-ray examination using iodinated contrast agents, and not resumed until 48 hours after it, provided that during the examination, renal function was found to be normal.
Other precautions:
- Patients are advised to continue to follow a diet with even carbohydrate intake throughout the day. Overweight patients are recommended to continue to follow a hypocaloric diet (but not less than 1000 kcal/day).
— It is recommended that routine laboratory tests be performed regularly to monitor diabetes mellitus.
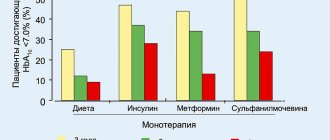
— Metformin in monotherapy does not cause hypoglycemia, however, it is recommended to exercise caution when using it in combination with insulin or other hypoglycemic agents (for example, sulfonylurea derivatives, repaglinide, etc.).
The use of Metformin is recommended for the prevention of type 2 diabetes mellitus in persons with prediabetes and additional risk factors for the development of overt type 2 diabetes mellitus, such as:
— Age less than 60 years;
— Body mass index (BMI) ≥ 30kg/m:;
- History of gestational diabetes mellitus;
— Family history of diabetes mellitus in first-degree relatives;
- Increased concentration of HDL cholesterol;
- arterial hypertension.
— Metformin did not affect the fertility of male and female rats when administered at doses three times the maximum recommended daily dose for humans.