Pharmacological properties of the drug Vitamin E-zentiva
Alpha tocopherol is the most biologically active of the tocopherols, stable in the form of acetate. In the body, it increases the resistance of cell membranes to damaging factors. Alpha-tocopherol enters the blood mainly with lymph; the maximum concentration of the drug in the blood serum is reached 4–8 hours after oral administration and returns to its original level within 24 hours. Alpha tocopherol is excreted in bile. The concentration of alpha-tocopherol in bile is stable and practically does not change with sharp fluctuations in the concentration of vitamin E in the blood plasma.
Indications for use of the drug Vitamin E-zentiva
Prevention of disorders of embryonic development of the fetus, auxiliary therapy of menstrual disorders. The drug is used as an adjuvant in the treatment of diseases of the respiratory tract, digestive tract, muscular system, joint apparatus, neurological diseases, dysfunction of the endocrine glands, malnutrition, diseases of the skin, gums, liver, anemia and perceptual hearing impairment. The drug is used during pregnancy and lactation.
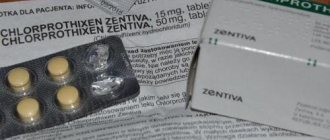
Chlorprothixene sanofi 50 mg 50 pcs. film-coated tablets
pharmachologic effect
Antipsychotic drug (neuroleptic), thioxanthene derivative.
It has antipsychotic, antidepressant, sedative, antiemetic effects, and has alpha-adrenergic blocking activity. It is believed that the antipsychotic effect is associated with blockade of postsynaptic dopamine receptors in the brain. The antiemetic effect is associated with blockade of the chemoreceptor trigger zone of the medulla oblongata. The sedative effect is due to an indirect weakening of the activity of the reticular system of the brain stem. Suppresses the release of most hormones of the hypothalamus and pituitary gland. However, as a result of blockade of the prolactin inhibitory factor, which inhibits the release of prolactin from the pituitary gland, the concentration of prolactin increases.
The chemical structure and pharmacological properties of thioxanthenes are similar to piperazine derivatives of phenothiazine.
Composition and release form Sanofi chlorprothixene 50 mg 50 pcs. film-coated tablets
Tablets - 1 tablet:
- Active substance: chlorprothixene hydrochloride 50 mg.
- Excipients: corn starch, lactose monohydrate, sucrose, calcium stearate, talc.
- Film shell composition: hypromellose 2910/5, macrogol 6000, macrogol 300, talc, aluminum varnish based on sunset yellow dye (E110).
10 pieces. - blisters (5) - cardboard packs.
Description of the dosage form
Orange film-coated tablets, round, biconvex; fracture appearance - the core is white to almost white.
Directions for use and doses
Individual. For oral administration for adults, the daily dose varies from 10 mg to 600 mg, for children - from 5 mg to 200 mg. The frequency of administration and duration of treatment are determined by the indications.
Pharmacokinetics
Metabolized in the liver. It is excreted primarily by the kidneys.
Indications for use Chlorprothixene sanofi 50 mg 50 pcs. film-coated tablets
Psychoses and psychotic states, accompanied by anxiety, fear, psychomotor agitation, aggressiveness, incl. for depressive-paranoid, circular schizophrenia, for simple sluggish schizophrenia with psychopath-like and neurosis-like symptoms and for other mental illnesses; discirculatory encephalopathy, traumatic brain injury (as part of combination therapy), alcoholic delirium; sleep disorders due to somatic diseases; the need for long-term therapy for states of excitation and anxiety, psychosomatic, neurotic and behavioral disorders in children; convulsive cough, spastic conditions in the gastrointestinal tract; premedication; dermatoses accompanied by persistent itching; allergic reactions.
Contraindications
CNS depression, incl. in case of intoxication with alcohol, barbiturates and other drugs that have a depressing effect on the central nervous system, pathological changes in the blood picture, myelodepression, pregnancy, lactation, hypersensitivity to chlorprothixene.
Application of Chlorprothixene sanofi 50 mg 50 pcs. film-coated tablets during pregnancy and breastfeeding
Contraindicated for use during pregnancy and lactation.
special instructions
It should not be used for epilepsy, a tendency to collapse, parkinsonism, heart defects in the stage of decompensation, tachycardia, cerebral atherosclerosis, severe liver and kidney dysfunction, hematopoietic disorders, cachexia, and in old age.
If it is necessary to use chlorprothixene, the risks and benefits of treatment should be compared in patients with chronic alcoholism, diseases of the cardiovascular system (increased risk of developing transient arterial hypotension), Reye's syndrome, as well as glaucoma or a predisposition to it, peptic ulcer of the stomach and duodenum, delayed urine, Parkinson's disease, epileptic seizures, hypersensitivity to other thioxanthenes or phenothiazines.
When using chlorprothixene, false-positive results of an immunological pregnancy test using urine, as well as false-positive results of a urine test for bilirubin, are possible.
During the treatment period, avoid drinking alcohol.
Impact on the ability to drive vehicles and operate machinery
During the treatment period, you should refrain from engaging in potentially hazardous activities that require increased attention and rapid psychomotor reactions.
Side effects Chlorprothixene sanofi 50 mg 50 pcs. film-coated tablets
From the side of the central nervous system: possible psychomotor inhibition, mild extrapyramidal syndrome, increased fatigue, dizziness; in isolated cases, a paradoxical increase in anxiety is possible, especially in patients with mania or schizophrenia.
From the digestive system: cholestatic jaundice is possible.
From the cardiovascular system: possible tachycardia, ECG changes, orthostatic hypotension.
From the organ of vision: possible clouding of the cornea and lens with visual impairment.
From the hematopoietic system: possible agranulocytosis, leukocytosis, leukopenia, hemolytic anemia.
From the endocrine system: frequent hot flashes, amenorrhea, galactorrhea, gynecomastia, weakened potency and libido are possible.
On the metabolic side: increased sweating, impaired carbohydrate metabolism, increased appetite with increased body weight are possible.
Dermatological reactions: photosensitivity, photodermatitis are possible.
Effects due to anticholinergic action: dry mouth, constipation, accommodation disturbances, dysuria.
Drug interactions
When used simultaneously with anesthetics, opioid analgesics, sedatives, hypnotics, antipsychotics, ethanol, ethanol-containing drugs, the inhibitory effect on the central nervous system is enhanced.
When used simultaneously with antihypertensive drugs, the hypotensive effect is enhanced.
When used simultaneously with anticholinergic, antihistamine, antiparkinsonian drugs, the anticholinergic effect is enhanced.
When used simultaneously with drugs that cause extrapyramidal reactions, an increase in the frequency and severity of extrapyramidal reactions is possible; with levodopa - the antiparkinsonian effect of levodopa may be inhibited; with lithium carbonate - severe extrapyramidal symptoms and neurotoxic effects are possible.
When used concomitantly with epinephrine, it is possible to block the alpha-adrenergic effects of epinephrine and, as a result, develop severe arterial hypotension and tachycardia.
When used simultaneously with phenothiazines, metoclopramide, haloperidol, reserpine, the development of extrapyramidal disorders is possible; with quinidine - an increased inhibitory effect on the heart is possible.
Use of the drug Vitamin E-Zentiva
The dose is selected individually, depending on the nature of the disease:
- infertility, spontaneous abortion: 200–300 mg daily; for menstrual irregularities (in combination with hormonal therapy) 300–400 mg every other day, starting from the 17th day of the cycle for 5 cycles; for menopausal autonomic disorders - 100 mg 3 times a day;
- for menstrual irregularities in juveniles before starting hormonal therapy - 100 mg 1-2 times a day for 2-3 months;
- for rheumatoid diseases - 100–300 mg daily for several weeks;
- for neurasthenia, fatigue - 100 mg daily for 30–60 days;
- for primary muscular dystrophy - up to 2000 mg per day; for other neurological indications, 300 mg per day for 30–60 days is sufficient;
- for cardiovascular diseases - 100 mg daily for 20–40 days;
- for nutritional anemia - 300 mg per day for 10 days;
- for chronic hepatitis with periods of exacerbation, 300 mg is prescribed daily for several months;
- for endocrine diseases - 300–500 mg daily;
- for periodontopathies - 200–300 mg per day;
- for induratio penis plastica - 300–400 mg daily for several weeks, then 100 mg daily for several months;
- for skin diseases - 100–200 mg per day for 20–40 days;
- as a component of antioxidant therapy, 400 mg is used 1–2 times a day;
- for prophylactic purposes, 100 mg is prescribed 1–2 times a day for 1–3 weeks.
Children over 12 years of age are prescribed 200–400 mg daily. The capsule is swallowed without chewing, washed down with a sufficient amount of water.
Chlorprothixene
When using any antipsychotic, there is a risk of developing neuroleptic malignant syndrome (hyperthermia, muscle rigidity, fluctuation of consciousness, instability of the autonomic nervous system). Patients with pre-existing psychoorganic syndrome, mental retardation, as well as those who abuse opiates and alcohol make up a significant proportion of deaths.
Treatment: discontinuation of the antipsychotic. Symptomatic therapy and general supportive treatment measures. Dantrolene and bromocriptine may be effective. After taking antipsychotics by mouth, symptoms may persist for more than one week.
Patients with rare conditions such as a small anterior chamber and a narrow angle may experience acute attacks of glaucoma due to pupil dilation.
Due to the risk of developing malignant arrhythmias, chlorprothixene should be used with caution in patients with a history of cardiovascular disease and in patients with a family history of long QT interval.
ECG monitoring is necessary before starting treatment. Chlorprothixene is contraindicated if the QTc interval at baseline is more than 450 ms in men and 470 ms in women (see section "Contraindications").
During therapy, the need for ECG monitoring should be assessed by the physician, taking into account the individual characteristics of the patient. During treatment, reduce the dosage if the QT interval is prolonged or discontinue therapy if the QTc is > 500 ms.
During treatment, periodic assessment of electrolyte balance is recommended.
The simultaneous use of other antipsychotic drugs should be avoided (see section "Interaction with other drugs").
Like other antipsychotics, chlorprothixene should be used with caution in patients with psychoorganic syndrome, seizures, liver, kidney and cardiovascular diseases in the later stages, as well as patients with myasthenia gravis and benign prostatic hyperplasia.
Caution must be exercised when using the drug in patients with
- pheochromocytoma,
- prolactin-dependent tumors,
- severe arterial hypotension or orthostatic dysregulation,
- Parkinson's disease,
- diseases of the hematopoietic system,
- hyperthyroidism,
- urinary problems, urinary retention,
- pyloric stenosis (pyloric stenosis), intestinal obstruction.
Like other psychotropic drugs, chlorprothixene may change the concentration of insulin and glucose in the blood, so patients with diabetes may require dosage adjustments of antidiabetic drugs.
Patients undergoing long-term treatment, especially with high doses, are subject to careful monitoring over time with periodic assessment of the need to reduce the maintenance dose.
Cases of venous thromboembolism (VTE) have been reported while taking antipsychotic drugs. Due to the fact that patients treated with antipsychotic drugs are often at risk for developing VTE, risk factors for VTE should be identified before and during treatment with chlorprothixene and precautions taken.
It has been reported that antipsychotics with α-adrenergic blocking effects may cause priapism; it is possible that chlorprothixene also has this property. If severe priapism occurs, medical intervention may be required.
Patients should be warned about the need to urgently seek medical help if objective and subjective signs of priapism appear.
Use of the drug in children and adolescents under 18 years of age
Chlorprothixene is not recommended for use in adolescent children. A sufficient number of studies have not been conducted to study the effectiveness and safety of the use of chlorprothixene for the treatment of children and adolescents.
Elderly patients
Cerebrovascular adverse reactions
In randomized placebo-controlled clinical trials of certain atypical antipsychotics in patients with dementia, a 3-fold increase in the risk of cerebrovascular adverse reactions was observed. The mechanism for this increased risk is unknown.
An increased risk cannot be ruled out when using other antipsychotics in other groups of patients.
Chlorprothixene should be used with caution in patients at risk of stroke. Elderly individuals are particularly at risk of developing orthostatic hypotension.
Increased mortality in older patients with dementia
Data from two large observational studies showed that older patients with dementia taking antipsychotic drugs had a nonsignificant increased risk of death compared with patients not taking antipsychotic drugs. There is no sufficient data to accurately assess the magnitude of the risk and the reasons for its increase. Chlorprothixene is not registered for the treatment of behavioral disorders in elderly patients with dementia.
Excipients
The tablets contain lactose monohydrate and sucrose. This drug should not be used in patients with rare hereditary problems such as lactose or fructose intolerance, lactase deficiency or glucose-galactose malabsorption, sucrase/isomaltase deficiency.
Interactions of the drug Vitamin E-Zentiva
Vitamin E facilitates the absorption and assimilation of retinol, preventing the development of vitamin A deficiency. At a dose exceeding 10 mg/kg body weight per day, vitamin E reduces the therapeutic effect of iron supplements in children with iron deficiency anemia. In low birth weight infants receiving iron supplements, vitamin E deficiency can cause hemolytic anemia. Vitamin E and its metabolites have an antagonistic effect on vitamin K. In patients taking oral anticoagulants for a long time, prolonged administration of vitamin E may increase the risk of bleeding. Vitamin E can increase the effectiveness of anticonvulsants in patients with epilepsy, in whose blood the concentration of lipid peroxidation products is increased.
Content
- Characteristics of chlorprothixene (tablet p/o 15 mg No. 50)
Composition: 1 tablet contains: chlorprothixene hydrochloride 15 mg.
Excipients: corn starch - 10 mg, lactose monohydrate - 92 mg, sucrose - 10 mg, calcium stearate - 1.
5 mg, talc - 1.
5 mg.
Film shell composition: hypromellose 2910/5 - 2.
011 mg, macrogol 6000 - 0.
069 mg, macrogol 300 - 0.
49 mg, talc - 1.
43 mg, aluminum varnish based on sunset yellow dye (E110) - 1 mg.
Orange film-coated tablets, round, biconvex.
The appearance of the fracture is that the core is white to almost white.
Pharmacological action: Antipsychotic drug (neuroleptic), thioxanthene derivative.
It has antipsychotic, antidepressant, sedative, antiemetic effects, and has alpha-adrenergic blocking activity.
It is believed that the antipsychotic effect is associated with blockade of postsynaptic dopamine receptors in the brain.
The antiemetic effect is associated with blockade of the chemoreceptor trigger zone of the medulla oblongata.
The sedative effect is due to an indirect weakening of the activity of the reticular system of the brain stem.
Suppresses the release of most hormones of the hypothalamus and pituitary gland.
However, as a result of blockade of the prolactin inhibitory factor, which inhibits the release of prolactin from the pituitary gland, the concentration of prolactin increases.
The chemical structure and pharmacological properties of thioxanthenes are similar to piperazine derivatives of phenothiazine.
Indications for Use: Psychoses and psychotic states accompanied by anxiety, fear, psychomotor agitation, aggressiveness, incl.
h.
with depressive-paranoid, circular schizophrenia, with simple sluggish schizophrenia with psychopathic and neurosis-like symptoms and with other mental illnesses; - dyscirculatory encephalopathy, traumatic brain injury (as part of combination therapy), delirium alcohol; - sleep disturbances in somatic diseases; - necessity long-term therapy of states of excitement and anxiety, psychosomatic, neurotic and behavioral disorders in children; - convulsive cough, spastic conditions in the gastrointestinal tract; - premedication; - dermatoses accompanied by persistent itching; - allergic reactions.
Method of Application: Individual.
For oral administration for adults, the daily dose varies from 10 mg to 600 mg, for children - from 5 mg to 200 mg.
The frequency of administration and duration of treatment are determined by the indications.
Interaction: When used simultaneously with anesthetics, opioid analgesics, sedatives, hypnotics, antipsychotics, ethanol, ethanol-containing drugs, the inhibitory effect on the central nervous system is enhanced.
When used simultaneously with antihypertensive drugs, the hypotensive effect is enhanced.
When used simultaneously with anticholinergic, antihistamine, antiparkinsonian drugs, the anticholinergic effect is enhanced.
When used simultaneously with drugs that cause extrapyramidal reactions, an increase in the frequency and severity of extrapyramidal reactions is possible; with levodopa - the antiparkinsonian effect of levodopa may be inhibited; with lithium carbonate - severe extrapyramidal symptoms and neurotoxic effects are possible.
When used concomitantly with epinephrine, it is possible to block the alpha-adrenergic effects of epinephrine and, as a result, develop severe arterial hypotension and tachycardia.
When used simultaneously with phenothiazines, metoclopramide, haloperidol, reserpine, the development of extrapyramidal disorders is possible; with quinidine - an increased inhibitory effect on the heart is possible.
Side effects: From the central nervous system: - possible psychomotor inhibition, mild extrapyramidal syndrome, increased fatigue, dizziness; - in isolated cases, a paradoxical increase in anxiety is possible, especially in patients with mania or schizophrenia.
From the digestive system: cholestatic jaundice is possible.
From the cardiovascular system: possible tachycardia, ECG changes, orthostatic hypotension.
From the organ of vision: possible clouding of the cornea and lens with visual impairment.
From the hematopoietic system: possible agranulocytosis, leukocytosis, leukopenia, hemolytic anemia.
From the endocrine system: frequent hot flashes, amenorrhea, galactorrhea, gynecomastia, weakened potency and libido are possible.
On the metabolic side: increased sweating, impaired carbohydrate metabolism, increased appetite with increased body weight are possible.
Dermatological reactions: photosensitivity, photodermatitis are possible.
Effects due to anticholinergic action: dry mouth, constipation, accommodation disturbances, dysuria.
Contraindications: CNS depression, incl.
h.
in case of intoxication with alcohol, barbiturates and other drugs that have a depressing effect on the central nervous system, pathological changes in the blood picture, myelodepression, pregnancy, lactation, hypersensitivity to chlorprothixene.
Should not be used in old age.
Overdose: In case of overdose, the following symptoms may occur: hyper- or hypothermia, convulsions, extrapyramidal symptoms, drowsiness, shock or coma.
In case of an overdose of Chlorprothixene, supportive and symptomatic treatment is provided.
Gastric lavage should be done as soon as possible; taking a sorbent is also recommended.
It is necessary to take measures aimed at maintaining the activity of the cardiovascular and respiratory systems.
Adrenaline should not be used as it may cause a decrease in blood pressure.
Extrapyramidal disorders can be treated with Biperiden, and seizures with Diazepam.
Special Instructions: Should not be used for epilepsy, a tendency to collapse, parkinsonism, heart defects in the stage of decompensation, tachycardia, cerebral atherosclerosis, severe liver and kidney dysfunction, hematopoietic disorders, cachexia, in old age.
If it is necessary to use chlorprothixene, the risks and benefits of treatment should be compared in patients with chronic alcoholism, diseases of the cardiovascular system (increased risk of developing transient arterial hypotension), Reye's syndrome, as well as glaucoma or a predisposition to it, peptic ulcer of the stomach and duodenum, delayed urine, Parkinson's disease, epileptic seizures, hypersensitivity to other thioxanthenes or phenothiazines.
When using chlorprothixene, false-positive results of an immunological pregnancy test using urine, as well as false-positive results of a urine test for bilirubin, are possible.
During the treatment period, avoid drinking alcohol.
Impact on the ability to drive vehicles and operate machinery During the treatment period, you should refrain from engaging in potentially hazardous activities that require increased attention and rapid psychomotor reactions.
Overdose of the drug Vitamin E-Zentiva, symptoms and treatment
When taken orally at the recommended dose, it does not manifest itself clinically. When taking the drug in a high dose (over 400 mg per day for a long time), the following are possible: dyspepsia, feeling of fatigue, general weakness, headache. Also noted are creatinuria, increased activity of creatine kinase, increased concentrations of cholesterol, TG, decreased concentrations of thyroxine and triiodothyronine in the blood serum, increased concentrations of estrogens and androgens in the urine. There is no specific antidote, treatment is symptomatic.