Instructions for use Temodal 140 mg 5 pcs. capsules
Composition and release form
Capsules 1 capsule:
- Active substance: temozolomide - 5 mg / 20 mg / 100 mg / 250 mg;
- Excipients: lactose anhydrous; colloidal silicon dioxide; sodium carboxymethyl starch; wine acid; stearic acid;
- Capsule shell: titanium dioxide; sodium lauryl sulfate; gelatin.
In dark glass bottles of 5 or 20 pcs.;
1 bottle in a cardboard pack. Capsules 1 capsule:
- Active substance: temozolomide - 140 mg / 180 mg;
- Excipients: lactose; sodium carboxymethyl starch; colloidal silicon dioxide; wine acid; stearic acid;
- Composition of capsule shell (140 mg): titanium dioxide; indigo carmine; sodium lauryl sulfate; gelatin;
- Composition of capsule shell (180 mg): titanium dioxide; iron oxide yellow dye; iron oxide red dye; sodium lauryl sulfate; gelatin;
- Ink composition for writing on the shell (black): shellac, ethanol; isopropanol; butanol; propylene glycol; purified water; ammonia, aqueous; potassium hydroxide; iron oxide dye black.
In dark glass bottles of 5 or 20 pcs.; 1 bottle in a cardboard pack.
Lyophilisate for preparing solution for infusion - 1 vial:
- Active substance: temozolomide - 100 mg;
- Excipients: mannitol - 600 mg, threonine - 160 mg, polysorbate 80 - 120 mg, sodium citrate dihydrate - 235.2 mg, concentrated hydrochloric acid - 160 mg.
Colorless glass bottles with a capacity of 100 ml, cardboard packs.
Composition and release form
Capsules 1 capsule:
- Active substance: temozolomide - 5 mg / 20 mg / 100 mg / 250 mg;
- Excipients: lactose anhydrous; colloidal silicon dioxide; sodium carboxymethyl starch; wine acid; stearic acid;
- Capsule shell: titanium dioxide; sodium lauryl sulfate; gelatin.
In dark glass bottles of 5 or 20 pcs.; 1 bottle in a cardboard pack.
Capsules 1 capsule:
- Active substance: temozolomide - 140 mg / 180 mg;
- Excipients: lactose; sodium carboxymethyl starch; colloidal silicon dioxide; wine acid; stearic acid;
- Composition of capsule shell (140 mg): titanium dioxide; indigo carmine; sodium lauryl sulfate; gelatin;
- Composition of capsule shell (180 mg): titanium dioxide; iron oxide yellow dye; iron oxide red dye; sodium lauryl sulfate; gelatin;
- Ink composition for writing on the shell (black): shellac, ethanol; isopropanol; butanol; propylene glycol; purified water; ammonia, aqueous; potassium hydroxide; iron oxide dye black.
In dark glass bottles of 5 or 20 pcs.; 1 bottle in a cardboard pack.
Lyophilisate for preparing solution for infusion - 1 vial:
- Active substance: temozolomide - 100 mg;
- Excipients: mannitol - 600 mg, threonine - 160 mg, polysorbate 80 - 120 mg, sodium citrate dihydrate - 235.2 mg, concentrated hydrochloric acid - 160 mg.
Colorless glass bottles with a capacity of 100 ml, cardboard packs.
Price for Temodal in Moscow. Buy Temodal at REDapteka.ru Instructions for use for Temodal.
Description of the dosage form
Capsules 5 mg: size No. 3 with an opaque green cap and a white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “5 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
Capsules 20 mg: size No. 2 with an opaque yellow cap and a white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “20 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
Capsules 100 mg: size No. 1 with an opaque pink cap and a white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “100 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
Capsules 140 mg: size No. 0, with an opaque blue cap and a white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “140 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
Capsules 180 mg: size No. 0, with an opaque orange cap and a white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “180 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
Capsules 250 mg: size No. 0 with opaque cap and white body. The capsules bear the following inscriptions in black ink: “Temodal” on the cap, “250 mg” on the body, a trademark in the form of stylized letters “SP” and two stripes.
All capsules contain white to light pink or light yellow-brown powder.
Lyophilisate for the preparation of a solution for infusion, white with a pinkish tint, containing no mechanical inclusions.
Characteristic
Temodal® is an imidazotetrazine alkylating drug with antitumor activity.
pharmachologic effect
Alkylating, antitumor.
Pharmacokinetics
Temodal® after oral administration is quickly absorbed and just as quickly excreted from the body in the urine. Temodal® quickly penetrates the BBB and enters the cerebrospinal fluid.
Cmax in plasma is achieved on average 0.5–1.5 hours (at the earliest - 20 minutes) after taking the drug. T1/2 from plasma is approximately 1.8 hours. Clearance, volume of distribution in plasma and T1/2 are not dose dependent.
Temodal® is weakly protein bound (10–20%). After oral administration of Temodal®, the average fecal excretion rate over 7 days was 0.8%, indicating complete absorption of the drug.
The main route of elimination of Temodal® is through the kidneys. 24 hours after oral administration, approximately 5–10% of the dose is determined unchanged in the urine; the remainder is excreted as 4-amino-5-imidazole-carboxamide hydrochloride (AIC), temozolomidic acid, or unidentified polar metabolites.
Taking Temodal® with food causes a decrease in Cmax by 33% and a decrease in AUC by 9%.
Plasma clearance of the drug is not affected by age, renal function, or tobacco use. The pharmacokinetic profile of the drug in patients with mild or moderate liver dysfunction is the same as in individuals with normal liver function.
The AUC is higher in children than in adults. The maximum tolerated dose (MTD) in children and adults was the same and amounted to 1000 mg/m2 per treatment cycle.
Pharmacodynamics
When released into the systemic circulation, at physiological pH values, it undergoes a rapid chemical transformation with the formation of the active compound - monomethyltriazenoimidazolecarboxamide (MTIC).
The cytotoxicity of MTIC is believed to be primarily due to alkylation of guanine at the O6 position and additional alkylation at the N7 position. Apparently, the cytotoxic damage resulting from this includes (triggers) the mechanism of aberrant restoration of the methyl residue.
Indications for use
Newly diagnosed glioblastoma multiforme was treated with combined radiation therapy followed by adjuvant monotherapy.
Malignant glioma (glioblastoma multiforme or anaplastic astrocytoma), in the presence of relapse or progression of the disease after standard therapy.
Advanced metastatic malignant melanoma - as a first-line therapeutic agent.
Contraindications for use
- hypersensitivity to temozolomide or other components of the drug, as well as to dacarbazine (DTIC);
- severe myelosuppression;
- pregnancy;
- lactation period;
- children under 3 years of age (recurrent or progressive malignant glioma) or up to 18 years of age (newly diagnosed glioblastoma multiforme or malignant melanoma);
- rare hereditary diseases such as galactose intolerance, lactase deficiency or glucose-galactose malabsorption.
Carefully:
- old age (over 70 years);
- severe renal or liver failure.
Use during pregnancy and children
No studies have been conducted on the use of Temodal® in pregnant women. During preclinical studies on rats and rabbits receiving Temodal® at a dose of 150 mg/m2, a teratogenic effect and toxic effects of the drug on the fetus were noted. In this regard, Temodal® is not recommended for use during pregnancy.
It is not known whether Temodal® is excreted in breast milk. During lactation, you should stop breastfeeding or take the drug.
Side effects
Newly diagnosed glioblastoma multiforme (adult patients). Table 1 shows the side effects observed in the treatment of patients with newly diagnosed glioblastoma multiforme during the combination and adjuvant phases of treatment in clinical trials (a causal relationship between the drug and side effects has not been established). The distribution of the frequency of side effects is made in accordance with the following gradation: very often - more than 10%, often - more than 1% - less than 10%, sometimes - more than 0.1% - less than 1%.
| Body system | Response frequency | Nature of the reaction | |
| combined treatment phase (with radiation therapy) n=288 | adjuvant phase of treatment n=224 | ||
| Mechanisms of resistance to infections | often | oral candidiasis, herpes simplex, pharyngitis, wound infection, other infection | oral candidiasis, other infection |
| Sometimes | herpes simplex, herpes zoster, flu-like symptoms | ||
| Blood and lymphatic system | often | leukopenia, lymphopenia, neutropenia, thrombocytopenia | anemia, febrile neutropenia, leukopenia, thrombocytopenia |
| Sometimes | anemia, febrile neutropenia | lymphopenia, petechiae | |
| The cardiovascular system | often | swelling, incl. swelling of the legs, hemorrhages | swelling of the legs, hemorrhages, deep vein thrombosis |
| Sometimes | palpitations, increased blood pressure, cerebral hemorrhage | swelling, incl. peripheral, pulmonary embolism | |
| Respiratory system | often | cough, shortness of breath | cough, shortness of breath |
| Sometimes | pneumonia, upper respiratory tract infection, nasal congestion | pneumonia, upper respiratory tract infection, sinusitis, bronchitis | |
| Endocrine system | Sometimes | cushingoid | cushingoid |
| Nervous system | Often | headache | headache, cramps |
| often | anxiety, emotional lability, insomnia, dizziness, balance disorder, impaired concentration, confusion and depression, convulsions, memory impairment, neuropathy, paresthesia, drowsiness, speech disorder, tremor | anxiety, depression, emotional lability, insomnia, dizziness, imbalance, impaired concentration, confusion, speech disorder, hemiparesis, memory impairment, neurological disorders (unspecified), neuropathy, paresthesia, somnolence, tremor | |
| Sometimes | apathy, behavioral disorders, depression, hallucinations, perceptual disturbances, extrapyramidal disorders, gait disturbance, hemiparesis, hyperesthesia, hypoesthesia, neurological disorders (unspecified), status epilepticus, parosmia, thirst | hallucinations, amnesia, gait disturbance, hemiplegia, hyperesthesia, sensory disturbances | |
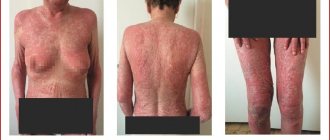
| Skin and subcutaneous tissue, mammary glands | Often | alopecia, rash | alopecia, rash |
| often | dermatitis, dry skin, erythema, skin itching, facial swelling | dry, itchy skin | |
| Sometimes | photosensitivity reactions, pigmentation disorders, exfoliation | erythema, pigmentation disorder, increased sweating, breast pain, facial swelling | |
| Musculoskeletal system | often | arthralgia, muscle weakness | arthralgia, musculoskeletal pain, myalgia, muscle weakness |
| Sometimes | back pain, musculoskeletal pain, myalgia, myopathy | back pain, myopathy | |
| Organs of vision | often | blurred vision | blurred vision, diplopia, limited visual fields |
| Sometimes | eye pain, hemianopsia, visual impairment, decreased visual acuity, limited visual fields | eye pain, dry eyes, decreased visual acuity | |
| Hearing organs and vestibular system | often | hearing impairment | hearing loss, ringing in the ears |
| Sometimes | ear pain, hyperacusis, tinnitus, otitis media | deafness, ear pain, dizziness | |
| Digestive system | Often | anorexia, constipation, nausea, vomiting | anorexia, constipation, nausea, vomiting |
| often | increased ALT activity, hyperglycemia, weight loss, abdominal pain, diarrhea, dyspepsia, dysphagia, stomatitis, taste disturbance | increased ALT activity, decreased body weight, diarrhea, dyspepsia, dysphagia, stomatitis, dry mouth, taste disturbance | |
| Sometimes | hypokalemia, increased alkaline phosphatase activity, weight gain, change in tongue color, increased activity of GGT, AST, liver enzymes | hyperglycemia, weight gain, bloating, fecal incontinence, hemorrhoids, gastroenteritis, dental diseases | |
| Genitourinary system | often | frequent urination, urinary incontinence | urinary incontinence |
| Sometimes | impotence | dysuria, amenorrhea, menorrhagia, vaginal bleeding, vaginitis | |
| Body as a whole | Often | fatigue | fatigue |
| often | fever, pain, radiation injury, allergic reaction | fever, pain, radiation injury, allergic reaction | |
| Sometimes | hot flashes, asthenia, deterioration, chills | asthenia, deterioration, chills | |
Laboratory indicators. Myelosuppression (neutropenia and thrombocytopenia) is a dose-limiting side effect. Among patients of both groups (with combination and adjuvant therapy), grade 3 and 4 changes in neutrophils, including neutropenia, were noted in 8% of cases, and in platelets, including thrombocytopenia, in 14% of cases.
Progressive or recurrent malignant glioma (adults and children over 3 years of age) or malignant melanoma (adults). The following adverse events noted when taking Temodal® are distributed according to the frequency of occurrence according to the following gradation: very often - ≥10% of cases, often - ≥1% - less than 10%, sometimes - ≥0.1% - less than 1% , rarely - 0.01% - less than 0.1% and very rarely - less than 0.01%.
From the hematopoietic system: very often - thrombocytopenia, neutropenia, lymphopenia; sometimes - pancytopenia, leukopenia, anemia. When treating patients with glioma and metastatic melanoma, cases of thrombocytopenia and neutropenia of the 3rd or 4th degree were observed in 19 and 17%, respectively, with glioma and in 20 and 22%, respectively, with melanoma. Hospitalization of the patient and/or discontinuation of Temodal® was required in 8 and 4% of cases, respectively, for glioma and in 3 and 1.3% for melanoma. Bone marrow suppression usually developed during the first few cycles of treatment, with a maximum between days 21 and 28; recovery usually occurred within 1–2 weeks. There were no signs of cumulative myelosuppression.
From the digestive system: very often - nausea, vomiting, constipation, anorexia; often - diarrhea, abdominal pain, dyspepsia, taste disturbances. The most common were nausea and vomiting. In most cases, these events were grade 1–2 (mild to moderate) and resolved spontaneously or were easily controlled with standard antiemetic therapy. The frequency of severe nausea and vomiting is 4%.
From the nervous system: very often - headache; often - drowsiness, dizziness, paresthesia, asthenia.
From the skin and skin appendages: often - rash, itching, alopecia, petechiae; very rarely - urticaria, exanthema, erythroderma, erythema multiforme, toxic epidermal necrolysis, Stevens-Johnson syndrome.
From the immune system: very rarely - allergic reactions, including anaphylaxis.
Other: very often - increased fatigue; often - weight loss, shortness of breath, increased body temperature, chills, general malaise; rarely - opportunistic infections, including pneumonia caused by Pneumocystis carinii; very rarely, the development of myelodysplastic syndrome (MDS) and secondary malignant processes, including leukemia, was observed, as well as the development of prolonged pancytopenia with the risk of developing aplastic anemia and irreversible infertility.
Drug interactions
Taking temozolomide together with ranitidine does not lead to a clinically significant change in the degree of absorption of temozolomide.
Co-administration with dexamethasone, prochlorperazine, phenytoin, carbamazepine, ondansetron, H2-histamine receptor antagonists or phenobarbital does not change the clearance of temozolomide.
Co-administration with valproic acid entails a mild but statistically significant decrease in the clearance of temozolomide.
No studies have been conducted to determine the effects of temozolomide on the metabolism and elimination of other drugs. Due to the fact that temozolomide is not metabolized in the liver and is weakly bound to proteins, its effect on the pharmacokinetics of other drugs is unlikely.
Use of temozolomide with other bone marrow depressants may increase the likelihood of myelosuppression.
Dosage
The method of administration and dosage regimen of a particular drug depend on its release form and other factors. The optimal dosage regimen is determined by the doctor. The compliance of the dosage form of a particular drug with the indications for use and dosage regimen should be strictly observed. Individual, depending on age and previous chemotherapy.
Orally, on an empty stomach, at least 1 hour before meals, without opening, without chewing, swallowing whole, with a glass of water. The prescribed dose should be taken using the minimum number of capsules possible.
Newly diagnosed glioblastoma multiforme (treatment of adult patients over 18 years of age).
Primary treatment is carried out in combination with radiation therapy. Temodal® is prescribed at a dose of 75 mg/m2 daily for 42 days simultaneously with radiation therapy (30 fractions in a total dose of 60 Gy). Dose reduction is not recommended, but the drug may be interrupted depending on tolerability. Resumption of the drug is possible throughout the entire 42-day period of combination treatment and up to 49 days, but only if all of the following conditions are met: absolute neutrophil count - not less than 1500 / μl, platelet count - not less than 100,000 / μl, general toxicity criterion (STS) - no higher than grade 1 (except for alopecia, nausea and vomiting). During treatment, blood tests should be performed weekly to count the number of cells.
Drug interactions Temodal
The combined use of Temodal with ranitidine or with food does not lead to clinically significant changes in the absorption of the drug. Co-administration of dexamethasone, prochlorperazine, phenytoin, carbamazepine, ondansetron, histamine H2 receptor antagonists or phenobarbital does not change the clearance of Temodal. Co-administration of valproic acid caused a mild but statistically significant decrease in the clearance of temozolomide. Use of Temodal with other bone marrow depressants may increase the likelihood of developing myelosuppression.
Temodal drug overdose, symptoms and treatment
Doses of 500, 750, 1000 and 1250 mg/m2 (total dose over a 5-day cycle) have been evaluated in patients clinically. Dose-related hematologic toxicity occurred at all doses but, as expected, was more severe at higher doses. One patient exceeded the dose of 2000 mg/day for 5 days, which resulted in pancytopenia, pyrexia, multiorgan failure and death. Patients taking recommended doses (150–200 mg/m2) for more than 5 days (up to 64 days) have been reported to develop bone marrow suppression (with or without infection), in some cases severe and prolonged, with death. In case of overdose, it is recommended to perform a hematological study and, if necessary, maintenance treatment.