Pharmacodynamics and pharmacokinetics
Pharmacodynamics
A hypnotic from the pyrazolo-pyrimidine group , it differs in chemical structure from benzodiazepines and other hypnotics. Selectively binds to type 1 benzodiazepine ω1 receptors.
Significantly reduces the time it takes to fall asleep, lengthens sleep time, and does not change the ratio of different phases of sleep. When used in recommended doses, it does not cause tolerance. It has a sedative , weak anxiolytic, anticonvulsant and systemic muscle relaxant effect.
Interacts with benzodiazepine receptors A-type GABA complexes . This leads to the opening of ionoform neuronal channels for chlorine molecules, the appearance of hyperpolarization and stimulation of inhibitory processes in the brain.
Pharmacokinetics
After consumption, about 71% of the dose taken is quickly absorbed from the intestines. The time for maximum concentration in the blood is one hour. Absolute bioavailability approaches 30%. Plasma concentrations are dose dependent. Eating slows down the time to reach the highest concentration without affecting the absorption of the drug.
Plasma protein binding by 60%. Able to be excreted in breast milk. Transformed in the liver to form inactive derivatives. The half-life of zaleplon is approximately 1 hour. Excreted in the form of inactive derivatives by the kidneys (71%) and through the intestines (17%).
Pharmacokinetics
Suction
After oral administration, it is quickly and almost completely (about 71%) absorbed from the gastrointestinal tract, Cmax in the blood is reached after 1 hour. As a result of first-pass metabolism, absolute bioavailability is 30%. Plasma concentration is directly proportional to dose. Taking the drug immediately after a meal can delay the time to reach Cmax by 2 hours without affecting the absorption of the drug.
Distribution
It is a fat-soluble compound. Vd after intravenous administration is 1.4±0.3 l/kg. Plasma protein binding is about 60% (the likelihood of interaction with other drugs is very low). Excreted in breast milk.
Metabolism
Aldehyde oxidase is involved in primary metabolism and leads to the formation of 5-oxozaleplon. CYP3A4 is also involved in the metabolism of zaleplon to desethyl zaleplon, which in turn is converted to 5-oxo-desethyl zaleplon by aldehyde oxidase. Subsequently, the oxidation products undergo conjugation with glucuronic acid. All metabolites are inactive. When used in doses up to 30 mg/day, no accumulation is observed. T1/2 of zaleplon - about 1 hour.
Removal
It is excreted as inactive metabolites, mainly in urine (71%) and feces (17%). Up to 57% of the dose taken is found in the urine in the form of 5-oxozaleplon or its metabolites, 9% of the dose in the form of 5-oxo-desetyl-zaleplon or its metabolites, and the remainder of the dose in the form of less significant metabolites. Among the metabolites excreted through the intestines, 5-oxozaleplon predominates. It is quickly eliminated from the body.
Pharmacokinetics in special clinical situations
Pharmacokinetics in elderly patients (including those over 75 years of age) do not differ significantly from those in younger patients.
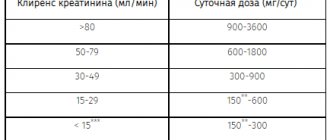
The pharmacokinetics of zaleplon in patients with renal failure do not differ significantly from those in healthy people, although their levels of inactive metabolites are higher.
Contraindications
- Sleep apnea.
- Severe forms of liver failure .
- Severe forms of respiratory failure .
- gravis .
- Pregnancy or lactation .
- Age less than 18 years.
- Sensitization to the components of the drug.
Andante should be prescribed with caution in case of renal or liver failure, chronic respiratory failure , tendency to drug addiction, alcoholism, depression.
Side effects
- Digestive reactions: vomiting, abdominal pain, nausea, diarrhea .
- Reactions from nervous activity: weakness, headache , drowsiness , anterograde amnesia , dizziness , depression , anxiety, aggressiveness, excitability, paresthesia , nightmares, attacks of rage, hallucinations , behavioral disorders, psychosis , the appearance of physical dependence even when used in therapeutic doses , withdrawal syndrome , the appearance of mental dependence, tremor, ataxia , irritability, changes in perception. of auto-aggression, epileptic seizures, hearing loss, depersonalization , and hypersensitivity to light, sound and other physical stimuli may appear
- Allergic reactions: skin rash , itching .
Andante
The patient should be warned about the short duration of the course of treatment and about the possibility of developing the syndrome after treatment.
The drug can be prescribed to elderly patients, incl. over 75 years old. The duration of treatment should be as short as possible and should not exceed 2 weeks. Treatment can be extended only after a thorough clinical examination of the patient.
Sleep disturbance can be the result of a physical or mental illness. If, after short-term treatment, sleep does not return to normal or the sleep disorder progresses, the diagnosis should be reconsidered.
If the patient wakes up shortly after midnight as a result of a short T1/2 Andante, it may be necessary to prescribe another drug with a longer T1/2. Patients should be warned about the need to take no more than 1 tablet per night.
Taking the drug for several weeks may be accompanied by a decrease in the hypnotic effect.
Taking the drug can lead to the development of physical and mental dependence, the likelihood of which is associated with taking large doses of the drug, long-term treatment, and the presence of alcohol and drug addiction.
When physical dependence has formed, abrupt withdrawal of the drug leads to the development of symptoms: headache, myalgia, severe anxiety, increased tension and irritability, psychomotor agitation, confusion. In severe cases, autoaggression, depersonalization, hearing loss, paresthesia in the extremities, increased reaction to light, sound and physical stimuli, hallucinations and epileptic seizures are possible.
Upon cessation of treatment, it is possible to develop recurrent insomnia or the appearance of transient and more pronounced symptoms of insomnia than at the beginning of treatment (syndrome). In this case, it is possible to develop other accompanying symptoms, incl. mood changes, anxiety, sleep disturbances or restlessness.
The development of anterograde amnesia and impairment of psychomotor functions is possible. To avoid the development of these symptoms, the drug should be taken only when the patient has the opportunity to sleep uninterruptedly for at least 4 hours after taking the drug.
Treatment should be discontinued if increased excitability, irritability, aggressiveness, disturbances in perception, nightmares, hallucinations, psychotic reactions and especially behavioral disorders occur. Children and elderly patients are most likely to develop such symptoms.
The drug is not intended for the treatment of depression and/or anxiety, because can be used to realize suicidal intentions, often accompanying depressive disorders. When treating patients with depression, a minimum dose of the drug should be prescribed to avoid deliberate overdose.
It is not recommended to prescribe the drug to patients with severe liver failure due to the risk of developing encephalopathy.
If you are lactose intolerant, please note that 5 mg of the drug contains 67 mg of lactose, 10 mg - 134 mg.
Due to the lack of data on use during pregnancy, use during pregnancy is not recommended. When prescribing the drug to women of childbearing age, the doctor should warn patients in each individual case about the need to immediately consult a doctor in case of conception or when planning pregnancy.
When using the drug in the third trimester of pregnancy or using large doses of the drug during childbirth, the newborn may develop hypothermia, muscle hypotension, and respiratory failure. Newborns whose mothers regularly took benzodiazepines in the last weeks of pregnancy may develop physical dependence and risk developing symptoms of the syndrome. Due to the penetration of the drug into breast milk, taking the drug during lactation is contraindicated.
During the treatment period, it is necessary to refrain from driving vehicles and engaging in other activities that require increased concentration and speed of psychomotor reactions.
Instructions for use Andante (Method and dosage)
The duration of therapy should not be more than two weeks. It is recommended to take the drug orally before bedtime and two hours after meals.
The recommended dose for adults is 10 mg. The highest daily dose is 10 mg. For elderly patients, Andante tablets are prescribed 5 mg (due to increased sensitivity to hypnotics).
For mild forms of liver failure, the daily dose is 5 mg (due to slow evacuation from the body).
For mild forms of renal failure, no dosage adjustment is required.
Overdose
An overdose does not cause life-threatening conditions if the drug is not taken together with other drugs that suppress nervous activity. In case of overdose, you should always remember the possibility of combined poisoning.
Signs of overdose: confusion, drowsiness leading to coma , lethargy, ataxia , respiratory failure, low blood pressure, fatal cases are rare.
Therapy: Flumazenil can be used as an antidote , gastric lavage in the first hours of administration, administration of activated charcoal , control of respiratory and cardiac activity.
Interaction
The simultaneous use of neuroleptics , other hypnotics, sedatives, anxiolytics, antiepileptics, antidepressants, antihistamines , anesthesia drugs , opioid-type analgesics leads to stimulation of the sedative effect of the drug.
When used together with opioid-type analgesics, it is possible to develop a euphoric effect from taking the latter, which can cause drug dependence.
When used together with Cimetidine, the content of zaleplon in the blood almost doubles.
When used simultaneously with selective CYP3A4 blockers ( Erythromycin, Ketoconazole ), the concentration of zaleplon in the blood also increases.
When used together with CYP3A4 stimulants (Carbamazepine, Rifampicin, phenobarbital-like drugs ), the effectiveness of zaleplon may be suppressed.
The drug does not affect the pharmacokinetics and pharmacodynamics of Warfarin and Digoxin .
Pharmacological properties of the drug Andante
Zaleplon has high selectivity and low affinity for type 1 benzodiazepine receptors. In patients with primary psychophysiological insomnia, depending on age, taking zaleplon at a dose of 5–10 mg reduces the latent time elapsed before falling asleep. Zaleplon prolongs the duration of sleep in the first half of the night, while the drug does not affect the percentage ratio between different phases of sleep. 2- and 4-week use in any of the doses was not accompanied by the development of tolerance to the drug. When taken orally, it is quickly and almost completely (about 71%) absorbed, reaching a maximum concentration in the blood serum after 1 hour. As a result of first-pass metabolism, absolute bioavailability is about 30%. The degree of binding to plasma proteins is about 60%. Passes into breast milk. Aldehyde oxidase is involved in primary metabolism, resulting in the formation of 5-oxozaleplon. CYP 3A4 is also involved in the metabolism of zaleplon to form desethyl zaleplon, which in turn is converted to 5-oxo-desethyl zaleplon by aldehyde oxidase. Subsequently, the oxidation products undergo conjugation with glucuronic acid. All zaleplon metabolites do not have pharmacological activity. As the dose increases, the concentration of zaleplon in the blood serum increases proportionally. When used in a daily dose of up to 30 mg, no accumulation was detected. The half-life of zaleplon is about 1 hour. Excretion occurs in the form of inactive metabolites, mainly in urine (71%) and feces (17%). 57% of the dose taken is detected in the urine in the form of 5-oxo-zaleplon or its metabolites, 9% of the dose is detected in the form of 5-oxo-desethyl-zaleplon or its metabolites. Among the metabolites detected in feces, 5-oxozaleplon predominates. It is quickly eliminated from the body.
special instructions
The patient should be warned that the drug does not serve as a means for long-term therapy and withdrawal syndrome may occur after stopping its use. The course of treatment should in no case exceed two weeks.
The drug is approved for use in elderly patients.
After stopping the use of benzodiazepine-like drugs, there is a possibility of temporary symptoms of insomnia , as well as other associated reactions (anxiety, mood changes, restlessness).
During the period of use of Andante, it is advised to refrain from operating mobile mechanisms.
Pregnancy and lactation
The drug is contraindicated during pregnancy and lactation (breastfeeding).
When prescribing the drug to women of childbearing age, patients should be warned about the need to immediately consult a doctor in case of conception or when planning pregnancy.
If zaleplon is used in the third trimester of pregnancy or when the drug is used in high doses during childbirth, a newborn may develop hypothermia, muscle hypotension, and moderate respiratory failure as a result of the pharmacological action of the drug. Infants whose mothers regularly took a benzodiazepine or a benzodiazepine-like drug during the last weeks of pregnancy may develop physical dependence and be at risk of developing withdrawal symptoms.
Analogs
Level 4 ATX code matches:
Sonnat
Relaxon
Ivadal
Sanval
Zolpidem
Somnol
Imovan
Zopiclone
Selofen, Zopiclone, Somnol, Sonnat, Sonovan and others.