Allegra tablets ppo 180 mg No. 10
Compound
Active substance: fexofenadine hydrochloride 180 mg.
Excipients: croscarmellose sodium - 36 mg, pregelatinized starch - 180 mg, microcrystalline cellulose - 199.5 mg, magnesium stearate - 4.5 mg.
Shell composition: hypromellose E-15 - 4.26 mg, hypromellose E-5 - 2.835 mg, povidone - 0.765 mg, titanium dioxide (E171) - 3.038 mg, colloidal silicon dioxide - 1.095 mg, macrogol 400 - 5.91 mg, iron dye oxide ( pink mixture: a mixture of red iron oxide (E172) and titanium dioxide (E171)) - 0.038 mg, iron oxide dye (yellow mixture: a mixture of yellow iron oxide (E172) and titanium dioxide (E171)) - 0.060 mg.
Pharmacokinetics
Suction and distribution
Fexofenadine is rapidly absorbed after oral administration, Tmax is approximately 1-3 hours. The average Cmax value when taking 120 mg/day is approximately 289 ng/ml, and when taking 180 mg/day is approximately 494 ng/ml.
The pharmacokinetics of single and course doses of fexofenadine (orally up to 120 mg 2 times a day) are linear. At a dose of 240 mg 2 times / day, there is a slightly greater than proportional (8.8%) increase in AUC, indicating that the pharmacokinetics of fexofenadine is almost linear in the dose range from 40 to 240 mg / day.
The binding of fexofenadine to plasma proteins is 60-70%.
Metabolism
Fexofenadine is slightly metabolized in and outside the liver, which is confirmed by the fact that it is the only substance detected in significant quantities in the urine and feces of humans and animals.
Removal
With a course of taking the drug, the elimination curve of fexofenadine from plasma decreases biexponentially, and the final T1/2 is 11-15 hours.
According to currently available data, most of the dose taken is excreted unchanged in the bile, and up to 10% is excreted in the urine.
Indications for use
- Seasonal allergic rhinitis (to reduce symptoms) - 120 mg tablets;
- chronic idiopathic urticaria (to reduce symptoms) - 180 mg tablets.
Contraindications
- Hypersensitivity to any of the components of the drug;
- pregnancy;
- lactation period;
- children's age up to 12 years.
Carefully:
- in patients with chronic renal and liver failure, as well as in elderly patients (lack of clinical experience in this category of patients);
- in patients with cardiovascular diseases, incl. history (antihistamines can cause palpitations and tachycardia).
Directions for use and doses
The drug is intended for oral administration.
The recommended dose of fexofenadine for seasonal allergic rhinitis for adults and children aged 12 years and older is 120 mg 1 time / day before meals.
The recommended dose of fexofenadine for chronic urticaria for adults and children aged 12 years and older is 180 mg 1 time / day before meals.
Patients at risk
Studies in special risk groups (elderly patients, patients with renal and liver failure) have shown that they do not require adjustment of the dosage regimen.
Storage conditions
The drug should be stored out of the reach of children at a temperature not exceeding 25°C.
Best before date
3 years. After the expiration date, the drug cannot be used.
special instructions
It is recommended that a minimum of 2 hours be allowed between taking fexofenadine and antacids containing aluminum or magnesium hydroxide.
Description
Antiallergic agent - H1-histamine receptor blocker.
Use in children
Contraindicated in children under 12 years of age.
Pharmacodynamics
Fexofenadine (a pharmacologically active metabolite of terfenadine) is an antihistamine with selective antagonistic activity at histamine H1 receptors without anticholinergic and alpha-blocking effects. In addition, fexofenadine does not have a sedative effect or other effects on the central nervous system.
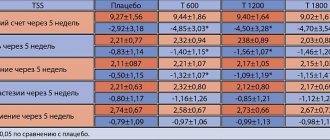
In human studies assessing histamine-induced wheals and flushing, the antihistamine effect of fexofenadine taken orally once or twice daily appears within 1 hour, peaks at 6 hours, and continues for 24 hours after administration. Even after 28 days of taking fexofenadine, no development of tolerance to the drug was detected. With a single oral dose of fexofenadine, a dose-dependent increase in the antihistamine effect is observed when the dose is increased from 10 mg to 130 mg. Using the same model of antihistamine action, it was found that a dose of at least 130 mg was required for continuous action over 24 hours. The maximum suppression of blistering and skin hyperemia is more than 80%.
In patients with seasonal allergic rhinitis who received up to 240 mg of fexofenadine 2 times a day for 2 weeks, the duration of the QTc interval did not differ from that when taking placebo.
There were also no changes in QTc when healthy volunteers took fexofenadine 60 mg 2 times / day for 6 months, 400 mg 2 times / day for 6.5 days and 240 mg / day for 1 year compared with the duration of QTc when taking placebo .
Even at plasma concentrations 32 times higher than therapeutic concentrations in humans, fexofenadine had no effect on delayed rectifier potassium channels in the human heart.
Side effects
In placebo-controlled clinical trials, the most commonly observed (≥1%, <10%) were:
headache (7.3%), drowsiness (2.3%), dizziness (1.5%) and nausea (1.5%). When taking fexofenadine, the incidence of the above adverse effects was similar to that when taking placebo.
In placebo-controlled studies with an incidence of less than 1% (the same for fexofenadine and placebo) and post-marketing use, the following were observed:
weakness, insomnia, nervousness and sleep disturbances or unusual dreams (paronyria), such as nightmares; tachycardia, palpitations; diarrhea.
In rare cases (≥0.01%, <0.1%):
exanthema, urticaria, itching and other hypersensitivity reactions, such as angioedema, difficulty breathing, shortness of breath, skin hyperemia, systemic anaphylactic reactions.
Use during pregnancy and breastfeeding
Pregnancy
There is insufficient data on the use of fexofenadine in pregnant women. Limited animal studies have shown no evidence of adverse effects on pregnancy, fetal development, childbirth or postnatal development. Fexofenadine should not be used during pregnancy.
Lactation
There are no data on the content of fexofenadine in breast milk when taken by women during breastfeeding. However, terfenadine has been shown to be excreted in breast milk in women. Therefore, the use of fexofenadine during breastfeeding is not recommended.
Interaction
When fexofenadine is co-administered with erythromycin or ketoconazole, the plasma concentration of fexofenadine increases 2-3 times, but this is not accompanied by a significant prolongation of the QTc interval. There were no significant differences in the incidence of adverse effects when using these drugs as monotherapy and as part of combination therapy. Animal studies have shown that the above-mentioned increase in plasma concentrations of fexofenadine is likely due to improved absorption of fexofenadine and a decrease in its biliary excretion or secretion into the gastrointestinal tract.
There is no interaction between fexofenadine and omeprazole.
Does not interact with drugs metabolized in the liver.
Taking antacids containing aluminum or magnesium 15 minutes before taking fexofenadine leads to a decrease in the bioavailability of the latter as a result, apparently, of binding in the gastrointestinal tract.
Overdose
Symptoms: dizziness, drowsiness and dry mouth. Healthy volunteers took single doses of up to 800 mg and course doses of up to 690 mg twice daily for 1 month or 240 mg twice daily for 1 year without any significant adverse effects compared with placebo. The maximum tolerated dose for fexofenadine has not been established.
Treatment: in case of overdose, it is recommended to perform gastric lavage, take activated charcoal, and, if necessary, symptomatic and supportive therapy. Hemodialysis is ineffective.
Impact on the ability to drive vehicles and operate machinery
When taking the drug, it is possible to perform work that requires a high concentration of attention and speed of psychomotor reactions (with the exception of patients who have a non-standard reaction). It is therefore recommended to check your individual response to fexofenadine before engaging in such activities.
Allegra tablets film coated. 120 mg, 10 pcs.
Manufacturer
Sanofi-Aventis, France
Compound
The film-coated tablets are light pink, oblong, biconvex, engraved “012” on one side and a stylized “e” on the other.
Excipients:
croscarmellose sodium - 24 mg,
pregelatinized starch - 120 mg,
microcrystalline cellulose - 133 mg,
magnesium stearate - 3 mg.
Shell composition:
hypromellose E-15 - 2.84 mg, hypromellose E-5 - 1.89 mg, povidone - 510 mcg, titanium dioxide (E171) - 2.025 mg, colloidal silicon dioxide - 730 mcg, macrogol 400 - 3.94 mg, pink mixture of iron oxide dye (iron red oxide (E172), titanium dioxide (E171)) - 250 mcg, yellow mixture of iron dye (yellow iron oxide (E172), titanium dioxide (E171)) - 40 mcg.
pharmachologic effect
Fexofenadine (a pharmacologically active metabolite of terfenadine) is an antihistamine with selective H1 receptor antagonist activity without anticholinergic and alpha-adrenergic receptor blocking effects. In addition, fexofenadine does not have a sedative effect or other effects on the central nervous system.
In human studies assessing histamine-induced wheals and flushing, the antihistamine effects of fexofenadine taken orally once or twice daily appeared within 1 hour, peaked at 6 hours, and continued for 24 hours after dosing.
Even after 28 days of taking fexofenadine, no development of tolerance to the drug was detected. With a single oral dose of fexofenadine, a dose-dependent increase in the antihistamine effect is observed when the dose is increased from 10 mg to 130 mg. Using the same model of antihistamine action, it was found that a dose of at least 130 mg was required for continuous action over 24 hours. The maximum suppression of blistering and skin hyperemia is more than 80%.
In patients with seasonal allergic rhinitis who received up to 240 mg of fexofenadine 2 times a day for 2 weeks, the duration of the QTc interval (QT corrected) did not differ from that when taking placebo.
There were also no changes in QTc when healthy volunteers took fexofenadine 60 mg 2 times a day for 6 months, 400 mg 2 times a day for 6.5 days and 240 mg per day for 1 year compared with the QTc duration when taking placebo .
Even at plasma concentrations 32 times higher than therapeutic concentrations in humans, fexofenadine had no effect on delayed rectifier potassium channels in the human heart.
Indications
— seasonal allergic rhinitis (to reduce symptoms) — tablets, 120 mg;
- chronic idiopathic urticaria (to reduce symptoms) - tablets, 180 mg.
Use during pregnancy and breastfeeding
Pregnancy
There is insufficient data on the use of fexofenadine in pregnant women. Limited animal studies have shown no evidence of adverse effects on pregnancy, fetal development, childbirth or postnatal development. Fexofenadine should not be used during pregnancy.
Lactation
There are no data on the content of fexofenadine in breast milk when taken by breastfeeding women. However, when taking terfenadine, it was observed to penetrate into the breast milk of lactating women. Therefore, the use of fexofenadine during breastfeeding is not recommended.
Contraindications
- hypersensitivity to any of the components of the drug;
- pregnancy;
- lactation period;
- children's age (up to 12 years).
Carefully:
- in patients with chronic renal and liver failure, as well as in elderly patients (lack of clinical experience in this category of patients);
- in patients with cardiovascular diseases, including a history (antihistamines can cause palpitations and tachycardia).
In case of impaired renal function.
Use with caution in chronic renal failure. No dosage regimen adjustment is required.
In case of liver dysfunction.
Use with caution in chronic liver failure. No dosage regimen adjustment is required.
Side effects
In placebo-controlled clinical trials, the most common (≥1%) were headache (7.3%), somnolence (2.3%), dizziness (1.5%), and nausea 1.5%. With fexofenadine, the incidence of the above adverse effects was similar to that with placebo.
In placebo-controlled studies, with an incidence of less than 1% (equal with fexofenadine and placebo) and post-marketing use of the drug, weakness, insomnia, nervousness, and sleep disturbances or unusual dreams (paroniria) such as nightmares were reported; tachycardia, palpitations; diarrhea.
Interaction
When fexofenadine is co-administered with erythromycin or ketoconazole, the plasma concentration of fexofenadine increases 2-3 times, but this is not associated with a significant prolongation of the QTc interval. There were no significant differences in the incidence of adverse effects when using these drugs in monotherapy and in combination. Animal studies have shown that the above-mentioned increase in plasma concentrations of fexofenadine is likely due to improved absorption of fexofenadine and a decrease in its biliary excretion or secretion into the gastrointestinal tract.
There is no interaction between fexofenadine and omeprazole.
Does not interact with drugs metabolized in the liver.
Taking antacids containing aluminum or magnesium 15 minutes before taking fexofenadine leads to a decrease in the bioavailability of the latter as a result of apparent binding in the gastrointestinal tract.
How to take, course of administration and dosage
The tablets are intended for oral administration.
The recommended dose of fexofenadine for seasonal allergic rhinitis for adults and children 12 years of age and older is 120 mg once a day before meals.
The recommended dose of fexofenadine for chronic urticaria for adults and children 12 years of age and older is 180 mg once daily before meals.
Patients at risk
Studies in special risk groups (elderly patients, patients with renal and liver failure) have shown that they do not require adjustment of the dosage regimen.
Overdose
Symptoms: dizziness, drowsiness and dry mouth. Healthy volunteers took single doses up to 800 mg, and course doses up to 690 mg 2 times a day for 1 month or 240 mg 2 times a day for 1 year without any significant adverse effects compared to placebo. The maximum tolerated dose for fexofenadine has not been established.
Treatment: in case of overdose, it is recommended to perform gastric lavage, take activated charcoal, and, if necessary, symptomatic and supportive therapy. Hemodialysis is ineffective.
Special instructions
It is recommended that a minimum of 2 hours be allowed between taking fexofenadine and antacids containing aluminum or magnesium hydroxide.
30 mg tablets are available for use in children aged 6 to 11 years.
Impact on the ability to drive vehicles and operate machinery
When taking the drug, it is possible to perform work that requires a high concentration of attention and speed of psychomotor reactions (with the exception of patients who have a non-standard reaction).
It is therefore recommended to check your individual response to fexofenadine before engaging in such activities.
Storage conditions
At a temperature not higher than 25°C. Keep out of the reach of children!
Best before date
3 years.
Active substance
Fexofenadine
Dosage form
pills
Purpose
For adults, Children over 12 years old
Indications
Allergy, Allergic rhinitis, Urticaria, Allergic conjunctivitis, Hay fever
Barcode and weight
Barcode: 3582910067061, 3582910069706 Weight: 0.018 kg