Treatment with Vivitrol is often indicated for people suffering from alcoholism. To overcome alcohol addiction, we implant the drug Vivitrol.
What is Vivitrol? Vivitrol is a brand name for naltrexone. It can be used to help people maintain abstinence from alcohol or opioids. Other naltrexone medications are available in oral tablet form, but Vivitrol is given as an intramuscular solution once a month. This is very convenient for people engaged in business or study. No need to take the drug daily.
How is Vivitrol treatment carried out? Possible complications and dangers. How is the maximum effect achieved when using the drug?
Vivitrol
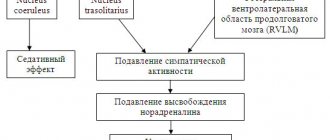
Vivitrol is an opioid antagonist that blocks the effects of alcohol consumption in the brain at the receptor level. It helps curb use, prevent binge relapses, and supports recovery from alcoholism. The Vivitrol treatment procedure involves local anesthesia. Before implantation, a test is performed to determine the patient’s tolerance to the active substance. This is necessary because the likelihood of an allergic reaction is very high. After administration of the drug into the body, the concentration of the active substance reaches a therapeutic level within 3 days. Subsequently, as the polymer matrix dissolves and diffuses, Vivitrol is gradually released over 30 days. After Vivitrol implantation, the patient experiences less pleasure from drinking alcohol. His craving for alcohol decreases or may disappear completely only after basic anti-alcohol treatment. Alcohol can create a feeling of power, strength and wit, a temporary refuge where one can escape pain, stress and fear - at least for a while. However, those who follow this path sooner or later learn that alcohol, which once provided them with pleasure and comfort, an escape from personal struggles, soon becomes the biggest struggle of their lives. However, quitting alcohol, which is cripplingly addictive, is not easy—even when drinking it disrupts relationships, family life, work, and personality. For people who have undergone treatment and decide to stop drinking alcohol, treatment with Vivitrol may reduce the risk of relapse by reducing cravings.
What is Vivitrol implantation?
The Vivitrol implant consists of small granules that are diluted in a special solvent and injected under the skin. The injected drug is slowly absorbed and is eliminated from the body for a long time. Implantation of Vivitrol is currently quite often used in Russia in combination with the main treatment of alcoholism. Alcohol withdrawal symptoms are not usually life-threatening, but they can be so unpleasant and distressing that they prevent many people from returning to sobriety. Although Vivitrol does not cure hangovers or withdrawal symptoms, it can make the recovery period easier. Although it is a useful tool in addiction therapy, it does not “cure” alcohol addiction, and the drug will not be effective unless the person has first received clinical treatment and is motivated to recover.
It is important that people undergoing addiction treatment first completely eliminate alcohol substitutes and then abstain from them for 7-10 days before starting Vivitrol; otherwise, this prescription drug may cause severe withdrawal symptoms.
After receiving the implant, a person still needs to see a doctor regularly and undergo ongoing therapy. This therapy usually consists of attending group or individual psychotherapy, which provides sustainable recovery. It will help you focus your energy on changing behavior and restoring your quality of life.
Vivitrol: instructions for use
Before using the drug, remove it from the refrigerator and leave it at room temperature to warm up. Then the doctor puts a needle on the syringe, takes about 4 ml from the bottle with the solvent, and then injects it into the bottle with the powder. Next, the suspension is mixed for one minute; it should acquire a milky white color with a yellowish tint. After this, approximately 4.2 ml of suspension is taken from the bottle, the needles are changed and injected deep into the gluteal muscle. Vivitrol instructions state that the drug must be administered 4 times a month, i.e. Once a week, deep into the annual muscle, with each new injection the muscles must be alternated. If the patient misses the next dose, it is recommended to give the injection as quickly as possible.
Vivitrol instructions for use also state that you cannot replace the solvent or inject the drug intravenously or subcutaneously, only deep into the gluteal muscle.
Contraindications:
- taking narcotic analgesics;
- hypersensitivity to one of the components of the drug;
- the patient is in a state of withdrawal syndrome (“withdrawal”), and in the case of alcohol, it is prohibited to use it for “delirium tremens”;
- hepatitis, liver dysfunction;
- state of alcohol intoxication, positive test for opioids in the urine, i.e. Before use, the patient must be sober and not use drugs for a long time.
- As for pregnancy and lactation, the effects of Vivitrol have not been studied here. It is believed that the drug should only be administered when the potential benefit to the mother outweighs the potential risk to the developing fetus.
Side effects during treatment with Vivitrol
- Diarrhea
- Stomach upset
- Headache
- Anxiety or nervousness
- Exhaustion or trouble sleeping
- Muscle or joint pain
Treatment with Vivitrol during pregnancy
Women who are pregnant or breastfeeding are generally advised not to use Vivitrol. Studies have been conducted in the United States on the safety and effectiveness of Vivitrol during pregnancy.
One study published in the 2002 issue of the Australian and New Zealand Journal of Obstetrics and Gynecology indicated that Vivitrol implantation was successful in combating alcohol dependence in pregnant women. The experiment involved 8 volunteers, pregnant women with alcoholism, and an Australian research center used Vivitrol implantation during pregnancy. Study participants were able to avoid drinking alcohol during pregnancy. Neither the women nor their babies had any adverse outcomes during or after childbirth.
The effect of the drug Vivitrol
Vivitrol prevents endorphins from binding to opioid receptors in people who drink alcohol. It directly blocks the binding of alcohol substitutes to opioid receptors in the brain. Treatment with Vivitrol can make alcohol addiction treatment much more effective by helping prevent relapse. People who use Vivitrol in combination with primary treatment achieve a 90 percent success rate, compared with 35 percent who received standard therapy. Those who used Vivitrol in conjunction with rehabilitation and ongoing therapy were 17 times less likely to relapse than those who did not take Vivitrol.
Although the use of Vivitrol is very successful in preventing relapses, it is necessary to completely complete a detoxification and mental state stabilization program before using this drug. It is very important to ensure that there are no toxic residues left in the body or signs of withdrawal before starting Vivitrol.
Treatment with Vivitrol?
Treatment with Vivitrol without prior combination therapy is still controversial and can sometimes be dangerous. As practice shows, the administration of Vivitrol without prior treatment does not give the desired effect. Moreover, quite often this is contraindicated. The maximum effect when using this drug is achieved only through complex therapy carried out at the Preobrazhenie clinic. If you need help finding a treatment center, you can contact us today at We will discuss Vivitrol treatment and other therapies with you. When it comes to planning treatment, it's wise to understand all your options so you can choose the best methods to get rid of the problem.
Possible long-term consequences of Vivitrol implantation
An Australian study using rats was conducted 10 years later in 2012. Research has shown the potential for potential changes in neurochemistry and behavior in the adult offspring of those who used Vivitrol implantation during pregnancy. The results showed that over-exposure to Vivitrol stimulated:
- A decrease in brain activity, especially in those areas that control motor activity, leads to the development of Parkinson's disease.
- Increased risk of taking alcohol and drugs.
- Hypersensitivity to morphine and its derivatives.
The use of Vivitrol implantation during pregnancy and lactation requires more detailed studies and observations of adults who were exposed to the drug in utero.
Vivitrol
Hepatotoxicity
Taking excessive doses of naltrexone can lead to hepatocellular disorders.
Naltrexone is contraindicated in acute hepatitis and liver failure. The prescription of Vivitrol to patients with acute liver disease should be carefully considered and justified, taking into account the risk of liver dysfunction. The ratio between the safe dose of naltrexone and the dose that causes liver damage is ≤ 5 times.
When used in recommended doses, Vivitrol is not hepatotoxic. Patients should be warned of the risk of liver problems and advised to seek medical attention if symptoms of hepatitis occur. If such symptoms occur, treatment with Vivitrol should be discontinued.
Eosinophilic pneumonia
During clinical studies, one case of incident and one case of suspected eosinophilic pneumonia was diagnosed. In both cases, the patients required hospitalization and were treated with antibiotics and glucocorticosteroids.
The possibility of developing eosinophilic pneumonia in a patient receiving Vivitrol should be considered, and patients should be advised to immediately seek medical help if symptoms such as progressive shortness of breath and hypoxia appear.
Clinicians should consider the possibility of eosinophilic pneumonia in patients who are resistant to antibiotics.
Hypersensitivity reactions
When taking the drug Vivitrol, side effects such as urticaria, angioedema, and anaphylaxis may develop. Patients should be warned that if hypersensitivity reactions develop, they should immediately consult a doctor and stop further treatment with the drug.
Opioid overdose after attempting to overcome opioid blockade
After opioid detoxification, patients typically have reduced tolerance to opioids. Vivitrol blocks the effects of exogenous opioids for 28 days, but there have been cases of fatal opioid overdose in patients who took opioids before receiving a new dose of Vivitrol or who missed their next Vivitrol injection.
Patients treated with Vivitrol may be sensitive to lower doses of opioids than before treatment. This can cause potentially life-threatening opioid toxicity (respiratory failure or respiratory arrest, collapse, etc.). Patients should be warned that they may become sensitive to lower doses of opioids after stopping treatment with Vivitrol.
It is important to be aware of decreased tolerance to opioids at the end of the dosing interval, which is approximately one month after the injection of the drug and if the next injection is missed. Patients and their families should be warned about opioid sensitivity and the risk of opioid overdose.
Although Vivitrol is a potent opioid receptor antagonist with long-lasting pharmacological effects, the blockade it causes can be overcome. Patients who attempt to overcome this blockade by administering large doses of exogenous opioids risk receiving a lethal dose of opioids and putting their lives in danger. The concentration of opioids in the plasma immediately after their administration may be sufficient to overcome the competitive blockade of opioid receptors, and as a result, the patient may immediately develop life-threatening opioid intoxication, manifested by respiratory depression and collapse. Patients should be aware of the serious consequences of attempting to overcome opioid receptor blockade.
The effect of the drug on the duration of contractions and childbirth
The effect of the drug on the duration of labor and labor is unknown.
Effect of the drug on laboratory blood parameters
In patients receiving Vivitrol treatment, an increase in eosinophil levels in the blood was noted, but after several months of treatment, eosinophil levels returned to normal.
In patients receiving high doses of the drug, there was a decrease in platelet counts by an average of 17.8 x 103 μl. However, randomized controlled trials have not proven the effect of Vivitrol on increasing bleeding.
Patients with opioid dependence treated with the drug for 24 weeks experienced a decrease in platelet counts of an average of 62.8 x 103 μl. In patients in the placebo group, platelet counts decreased by an average of 39.9 x 103 μl. However, randomized controlled trials have not demonstrated the effect of Vivitrol on increasing bleeding.
The increase in blood aspartate aminotransferase activity during treatment with Vivitrol was the same as during treatment with oral naltrexone and was 1.5% compared to 0.9% in the placebo group.
In clinical studies lasting up to 6 months involving patients with opioid dependence, 89% of patients were diagnosed with hepatitis C, and 41% of patients were diagnosed with HIV infection. Increases in liver enzyme activity (alanine aminotransferase, aspartate aminotransferase, and gamma-glutamyltransferase) were frequently observed in the study. These side effects were observed more frequently in the Vivitrol 380 mg group than in the placebo group.
Patients with alanine aminotransferase or aspartate aminotransferase activity greater than three times the upper limit of normal were not included in the study.
In patients treated with Vivitrol, transaminase levels greater than 3 times the upper limit of normal and requiring immediate treatment were observed more frequently than in patients receiving placebo. This increase in enzyme activity was observed in 20% of patients treated with Vivitrol, compared with 13% of patients in the placebo group.
Increases in aspartate aminotransferase activity greater than three times the upper limit of normal were also more common in Vivitrol-treated patients (14%) compared with placebo-treated patients (11%).
In patients with opioid dependence treated with Vivitrol, alanine aminotransferase activity increased by an average of 61 IU/L, in patients in the placebo group - by an average of 48 IU/L.
In patients with opioid dependence treated with Vivitrol, aspartate aminotransferase activity increased by an average of 40 IU/L, in patients in the placebo group - by an average of 31 IU/L.
In clinical studies, patients receiving Vivitrol experienced increases in creatine phosphokinase activity, typically 1-2 times the upper limit of normal. A similar increase in creatine phosphokinase activity is also observed during treatment with oral naltrexone. However, there are known cases of a 4-fold and even 35-fold increase in the activity of this enzyme in a group of patients receiving oral naltrexone and Vivitrol, respectively.
In 39% of patients with opioid dependence treated with Vivitrol, an increase in creatine phosphokinase activity above normal values was observed; in the group of patients receiving placebo, an increase in creatine phosphokinase activity was observed in 32% of patients.
In some cases, in patients receiving placebo, creatine phosphokinase activity increased by 41.8 times the upper limit of normal; in the group of patients treated with Vivitrol, there are known cases of increased creatine phosphokinase activity by 22.1 times compared to the upper limit of normal.
Some ELISA urine tests may produce false-positive results for some drugs, especially opioids. See specific test instructions for more information.
Warnings and Precautions
Removing Vivitrol blockade
In an emergency setting for patients receiving Vivitrol treatment, the preferred method of pain control is regional analgesia, benzodiazepine sedation, and the use of non-narcotic analgesics or general anesthesia.
In situations requiring the use of narcotic analgesia, the required dose of opioids may be higher than usual, resulting in more profound and prolonged respiratory depression.
It is preferable to use a fast-acting narcotic analgesic that reduces the duration of respiratory depression. The amount of analgesic administered should be determined according to the needs of the patient. Non-receptor-mediated effects (eg, facial swelling, pruritus, generalized erythema, or bronchospasm) may occur, presumably due to histamine secretion.
Regardless of the agent chosen to reverse the Vivitrol block, the patient should be closely monitored by appropriately qualified nursing staff in a specially equipped intensive care unit.
Depression and suicidal behavior
During controlled clinical trials of Vivitrol, adverse events of a suicidal nature (suicidal ideation, suicide attempts, completed suicides) were infrequent but occurred more frequently in the Vivitrol-treated group than in the placebo-treated group (1% vs. 0). In some cases, suicidal thoughts and behavior appeared in the patient after completion of the study, but were a consequence of depression that developed during drug treatment. There were two suicides, in both cases the patients were treated with Vivitrol.
Discontinuation due to depression occurred more frequently in the Vivitrol group (1%) than in the placebo group (0).
In a 24-week placebo-controlled study, adverse events related to depressive mood were observed in 10% of patients treated with Vivitrol 380 mg and in 5% of patients receiving placebo injections.
Patients with alcohol dependence, including patients receiving treatment with Vivitrol, should be closely monitored for signs of depression or suicidal ideation. Family members and caregivers of patients should be advised to monitor closely for symptoms of depression or suicidal behavior and to immediately report such symptoms to their healthcare provider.
Injection site reactions
Vivitrol injections may be accompanied by pain, tenderness, induration, swelling, erythema and itching. During clinical trials, one patient developed a lump that continued to enlarge 4 weeks after injection, followed by necrosis that required surgery to remove. Other cases of injection site reactions including abscess, sterile abscess, induration and necrosis have been observed during post-marketing surveillance, some of which required surgery to resolve. Patients should be warned about the need to inform their doctor about the occurrence of any reaction at the injection site.
Alcohol withdrawal
The use of Vivitrol not only does not exclude, but also does not reduce the manifestation of symptoms associated with the withdrawal of alcohol intake.
Retinal occlusion
Retinal artery occlusion after injection of another drug containing a copolymer of lactic acid and glycolic acid was very rare in post-marketing studies and occurred in the presence of an anomalous arteriovenous anastomosis. During clinical and post-marketing studies of Vivitrol, not a single case of retinal artery obstruction was observed. Vivitrol should be used as an injection into the gluteal muscle and avoid accidental injection into a blood vessel.
Are there any dangers of Vivitrol treatment?
As a rule, Vivitrol implants are a safe treatment in most cases. However, risks may be present and complications may include complications at the site of Vivitrol implantation
- Infection.
- Inflammation - induration, swelling, erythema, bruising, necrosis.
- Local allergic reaction - pain, redness, itching. tenderness, or itching.
Dangers of Vivitrol Treatment
Withdrawal symptoms: if alcohol metabolites are present in the body at the time of Vivitrol administration, the drug should absolutely not be used. Vivitrol blocks the effect of intoxication. Patients do not feel the effect of drinking alcohol. If they try to drink in small doses. However, it should be noted that drinking large doses of alcohol to try to bypass the blockade can lead to serious complications, coma or death. During treatment with Vivitrol, patients may not experience the expected effects of analgesics, antidiarrheals, or antitussives. Vivitrol may cause liver damage. Therefore, before starting treatment with Vivitrol, an examination is necessary. Patients should notify their doctor immediately if they develop symptoms and/or signs of liver disease. Treatment with Vivitrol can stimulate the formation of depression. If you experience the first signs of depression, you should immediately consult a doctor. Vivitrol can cause allergic pneumonia. Patients should notify their doctor immediately if they develop signs and symptoms of pneumonia, including shortness of breath, cough, or wheezing. Vivitrol has been shown to be most effective when used as an integrated therapy program that includes counseling and support.