Axamon solution d/im/sc injected 15 mg/ml 1 ml x10
ATX code: N07AA (Anticholinesterase drugs) Active substance: ipidacrine (ipidacrine) Rec.INN registered by WHO Dosage form AKSAMON solution for intramuscular and subcutaneous administration 15 mg/ml: 1 ml amp. 10 pieces. reg. No.: LP-002247 dated 09.25.13 - Valid Release form, composition and packaging Solution for intramuscular and subcutaneous administration in the form of a clear, colorless liquid.
1 ml ipidacrine hydrochloride (in the form of monohydrate) 15 mg
Excipients: hydrochloric acid - up to pH 3.0, water for injection - up to 1 ml.
1 ml - glass ampoules (5) - contour cell packaging made of polyvinyl chloride film (2) - cardboard packs.
Clinical and pharmacological group: Cholinesterase inhibitor Pharmaco-therapeutic group: Anticholinesterase drug Pharmacological action Information on the drug is available only to specialists. Login or Register Indications Diseases of the peripheral nervous system (neuritis, polyneuritis, polyneuropathy, polyradiculopathy), bulbar palsy and paresis.
In the recovery period for organic lesions of the central nervous system, accompanied by motor disorders.
Myasthenia, myasthenic syndrome.
Demyelinating diseases (as part of complex therapy).
Alzheimer's disease, senile dementia of the Alzheimer's type.
Functional disorders of the central nervous system (decreased memory, ability to concentrate, motivation, initiative, disorientation, emotional lability, etc.) with encephalopathy (traumatic, vascular and other origins), cerebrovascular accident, traumatic brain injury, cerebral dysfunction with learning disabilities children.
Weakness of labor.
Intestinal atony.
Intoxication with anticholinergic drugs.
ICD-10 codes ICD-10 code Indication F00 Dementia in Alzheimer's disease F07 Personality and behavioral disorders caused by disease, damage or dysfunction of the brain G30 Alzheimer's disease G60 Hereditary and idiopathic neuropathy G61 Inflammatory polyneuropathy G62.1 Alcoholic polyneuropathy G63.2 Diabetic polyneuropathy G70 .2 Congenital or acquired myasthenia M54.1 Radiculopathy M79.2 Neuralgia and neuritis, unspecified O62 Disturbances of labor [birth forces]
Dosage regimen: Orally, subcutaneously, intramuscularly. Single dose – 10-40 mg. The maximum dose is 200 mg/day. The frequency of administration and duration of treatment depend on the indications. Side effects From the digestive system: anorexia, hypersalivation, nausea, vomiting, increased peristalsis, diarrhea, jaundice.
From the side of the central nervous system: dizziness (after repeated use), ataxia.
Allergic reactions: skin rash, itching.
Other: manifestations of m-cholinergic stimulating effect - bronchospasm, bradycardia.
Contraindications for use Epilepsy, extrapyramidal disorders with hyperkinesis, angina pectoris, severe bradycardia, bronchial asthma, tendency to vestibular disorders, pregnancy, lactation, hypersensitivity to ipidacrine. Use during pregnancy and lactation Contraindicated during pregnancy and lactation. Special instructions: Use with caution in case of gastric ulcer, thyrotoxicosis, diseases of the cardiovascular system. It is necessary to take into account the ability of ipidacrine to increase uterine tone.
Impact on the ability to drive vehicles and operate machinery
It should not be used in patients whose activities involve driving vehicles and work that requires high concentration and speed of psychomotor reactions.
Aksamon 5mg/ml 1ml 10 pcs. injection
pharmachologic effect
Cholinesterase inhibitor.
Composition and release form Aksamon 5 mg/ml 1 ml 10 pcs. injection
Solution for intramuscular and subcutaneous administration - 1 ml:
- active substance: ipidacrine hydrochloride monohydrate (in terms of anhydrous) - 5/15 ml;
- excipients: 0.1 M hydrochloric acid solution - to pH 3; water for injection - up to 1 ml.
Solution for intramuscular and subcutaneous administration, 5 mg/ml, 15 mg/ml. 1 ml of the drug in an ampoule with a dot or break ring made of colorless glass. 5 amp. placed in a blister pack made of PVC film. 2 blister packs are placed in a cardboard pack.
Description of the dosage form
Transparent colorless liquid.
Directions for use and doses
Pills
Inside. Doses and duration of treatment are determined individually depending on the severity of the disease.
Diseases of the peripheral nervous system, myasthenia gravis, myasthenic syndrome. 10–20 mg 1–3 times a day. The course of treatment is from 1 to 2 months. If necessary, the course of treatment can be repeated several times with a break between courses of 1–2 months.
Alzheimer's disease, encephalopathy, recovery period of organic diseases of the central nervous system, accompanied by motor disorders, incl. bulbar palsy and paresis. The daily dose is selected individually, usually 10–20 mg 2–3 times a day.
Demyelinating lesions of the nervous system. The dose can be increased to 20–40 mg 5–6 times a day. The maximum daily dose is 200 mg.
If one of the doses is missed, then the usual dose of the drug is taken at the next dose.
Solution for intramuscular and subcutaneous administration
S/c, i/m. Doses and duration of treatment are determined individually depending on the severity of the disease.
Diseases of the peripheral nervous system. Mono- and polyneuropathy of various origins, polyradiculopathy - 5-15 mg 1-2 times a day, course 10-15 days (in severe cases - up to 30 days); then treatment is continued with the tablet form of the drug. Myasthenia and myasthenic syndrome - 15-30 mg 1-3 times a day with further transition to the tablet form. The general course of treatment is 1–2 months. If necessary, treatment can be repeated several times with a break between courses of 1–2 months.
Diseases of the central nervous system. Bulbar paralysis and paresis - 5–15 mg 1–2 times a day, course 10–15 days; then, if possible, switch to tablet form. The recovery period for organic lesions of the central nervous system, accompanied by motor disorders, is 10–15 mg IM 1–2 times a day, course up to 15 days; then, if possible, switch to tablet form.
Treatment and prevention of intestinal atony. The initial dose is 10–15 mg 1–2 times a day for 1–2 weeks.
Pharmacodynamics
Stimulating neuromuscular transmission, neurometabolic, anticholinesterase.
A reversible cholinesterase inhibitor, directly stimulates impulse transmission at the neuromuscular synapse and in the central nervous system due to blockade of membrane potassium channels. It enhances the effect on smooth muscles of not only acetylcholine, but also adrenaline, serotonin, histamine and oxytocin. Aksamon® has the following pharmacological effects:
- improves and stimulates impulse conduction in the nervous system and neuromuscular transmission;
- improves conductivity in the peripheral nervous system, impaired due to injury, inflammation, exposure to local anesthetics, some antibiotics, potassium chloride, etc.;
- enhances the contractility of smooth muscle organs under the influence of agonists of acetylcholine, adrenaline, serotonin, histamine and oxytocin receptors, with the exception of potassium chloride;
- improves memory, inhibits the progressive development of dementia.
Does not have teratogenic, embryotoxic, mutagenic, carcinogenic or immunotoxic effects. Does not affect the endocrine system.
Pharmacokinetics
Suction and distribution
When administered subcutaneously or intramuscularly, it is rapidly absorbed. Cmax in the blood is achieved 25-30 minutes after administration.
Binding to blood plasma proteins is 40-50%. The drug quickly enters the tissues.
Metabolism and excretion
Metabolized in the liver. The drug is excreted through the kidneys (mainly by tubular secretion and only 1/3 by glomerular filtration) and extrarenally (through the gastrointestinal tract). T1/2 of the drug Aksamon® when administered parenterally is 2-3 hours. After parenteral administration, 34.8% of the drug dose is excreted unchanged in the urine.
Indications for use Aksamon 5mg/ml 1ml 10 pcs. injection
- diseases of the peripheral nervous system: mono- and polyneuropathy, polyradiculopathy, myasthenia gravis and myasthenic syndrome of various etiologies;
- diseases of the central nervous system: bulbar paralysis and paresis, the recovery period of organic lesions of the central nervous system, accompanied by motor disorders;
- intestinal atony.
Contraindications
- epilepsy;
- extrapyramidal disorders with hyperkinesis;
- angina pectoris;
- severe bradycardia;
- bronchial asthma;
- mechanical obstruction of the intestine or urinary tract;
- vestibular disorders;
- peptic ulcer of the stomach or duodenum in the acute stage;
- children under 18 years of age (there is no systematic data on use in children);
- hypersensitivity to any of the components of the drug.
Prescribed with caution for gastric and duodenal ulcers, thyrotoxicosis, diseases of the cardiovascular system, a history of obstructive diseases of the respiratory system or acute respiratory diseases.
Application Axamon 5mg/ml 1ml 10 pcs. solution for injection during pregnancy and lactation
The use of the drug increases the tone of the uterus and can lead to premature birth, therefore ipidacrine is contraindicated during pregnancy.
There are no data on the use of the drug during breastfeeding.
Contraindicated in children under 18 years of age (there are no systematic data on use in children.
special instructions
Impact on the ability to drive vehicles and operate machinery
During treatment, you should refrain from driving a car, other vehicles and machinery, as well as from engaging in potentially hazardous activities that require increased concentration and speed of psychomotor reactions.
Overdose
Symptoms: decreased appetite, bronchospasm, lacrimation, increased sweating, constriction of the pupils, nystagmus, increased gastrointestinal peristalsis, spontaneous bowel movements and urination, vomiting, jaundice, bradycardia, intracardiac conduction disturbances, arrhythmias, decreased blood pressure, restlessness, anxiety, agitation, feelings of fear, ataxia, convulsions, coma, speech disorders, drowsiness and general weakness.
Treatment: use of m-anticholinergic drugs (including atropine, cyclodol, metacin), symptomatic therapy.
Side effects Aksamon 5mg/ml 1ml 10 pcs. injection
The frequency of adverse reactions that may occur during use of the drug is given in the following classification: very common (>1/10 prescriptions), frequent (1/10-1/100 prescriptions), infrequent (1/100-1/1000 prescriptions), rare (1/1000-1/10,000 prescriptions), very rare (
From the mental side: infrequent - drowsiness.
From the nervous system: infrequently - dizziness, headache, convulsions; unknown frequency - tremor.
From the organ of vision: unknown frequency - miosis.
From the side of the heart: frequent - palpitations, bradycardia; unknown frequency - chest pain.
From the respiratory system: infrequent - increased secretion of bronchial secretions.
From the gastrointestinal tract: frequent - drooling, nausea; infrequently - vomiting; rare - diarrhea, epigastric pain.
From the liver and biliary tract: unknown frequency - jaundice.
From the skin and subcutaneous tissues: infrequently - itching, rash.
General disorders and disorders at the injection site: frequent - increased sweating; infrequent - general weakness; unknown frequency - hypothermia.
Salivation and bradycardia can be reduced with m-anticholinergic drugs (including atropine, cyclodol, metacin).
If itching or rash occurs, stop taking the drug. In other cases, reduce the dose or interrupt the drug for a short time (for 1-2 days).
Drug interactions
Axamon® enhances the sedative effect in combination with drugs that depress the central nervous system.
The action and side effects are enhanced when used together with other cholinesterase inhibitors and m-cholinomimetic drugs.
In patients with myasthenia gravis, the risk of developing a cholinergic crisis increases if Axamon is used simultaneously with other cholinergic drugs.
The risk of developing bradycardia increases if beta-blockers were used before starting treatment with Axamon®.
Reduces the inhibitory effect on neuromuscular transmission and the conduction of excitation along peripheral nerves by local anesthetics, aminoglycosides, and potassium chloride.
Axamon® can be used in combination with nootropic drugs.
Alcohol increases the side effects of the drug.
Modern experience in the use of anticholinesterase drugs in neurology
Anticholinesterase drugs are used in neurological practice to activate regeneration and reinnervation processes in diseases of the peripheral and central nervous system.
For many years, the drug “Prozerin” has been widely used in medical practice, which has a pronounced effect on peripheral cholinergic synapses.
At the end of the 80s. In the 20th century, the drug “Amiridin” with a combined anticholinesterase effect was synthesized. Since 2002, the drug has been produced under the trade name Neuromidin.
The pharmacological effects of anticholinesterase drugs lead to increased action of acetylcholine. The choice of a drug from the group of anticholinesterase drugs is determined by its pharmacological properties, duration of action, activity, ability to penetrate tissue barriers, as well as side effects.
Proserin - N-(meta-dimethylcarbamoyloxyphenyl)-trimethylammonium methyl sulfate has strong reversible anticholinesterase activity. The basis of its mechanism of action is the inhibition of acetylcholinesterase, an enzyme that hydrolyzes the mediator acetylcholine, which is accompanied by a slowdown in the rate of its destruction, accumulation and enhancement of action in the area of cholinergic synapses with restoration of neuromuscular conduction.
Due to the presence of a quaternary ammonium group in the Proserin molecule, the drug poorly penetrates the blood-brain barrier and mainly affects peripheral cholinergic structures.
A significant difference between Neuromidin (ipidacrine) and Proserin is the ability of Neuromidin to block the potassium permeability of the membrane, which leads to an extension of the repolarization phase of its action potential and an increase in the activity of the presynaptic axon. This process is accompanied by an increase in the entry of calcium ions into the presynaptic terminal and, as a consequence, an increase in the release of the transmitter acetylcholine into the synaptic cleft in all synapses, contributing to increased stimulation of the postsynaptic cell.
In addition, Neuromidin has the effect of reversible inhibition of cholinesterase in cholinergic synapses, which further increases the accumulation of the neurotransmitter in the synaptic cleft and, accordingly, enhances the functional activity of the postsynaptic cell. However, the inhibition of the action of acetylcholinesterase in Neuromidin is less pronounced compared to Proserin.
Thus, Neuromidin stimulates the presynaptic nerve fiber, increases the release of the neurotransmitter into the synaptic cleft, reduces the destruction of the neurotransmitter acetylcholine by the enzyme, increases the activity of the postsynaptic cell by direct membrane and mediated mediator effects, that is, it acts on all parts of the conduction of excitation, in contrast to Proserin, which reduces metabolism mediator only at cholinergic synapses.
The M-cholinomimetic effects of Proserin and Neuromidin are similar, however, the activity of the effect on M-cholinergic receptors is higher in Proserin.
Neuromidin has the following pharmacological effects: restores and stimulates neuromuscular transmission, conducts impulses in the peripheral nervous system, disturbed due to the influence of various factors. Neuromidin increases muscle contractile activity under the influence of acetylcholine and other mediators: adrenaline, serotonin, histamine and oxytocin, having a direct effect on myofibrils, while Prozerin does not have this property.
Neuromidin, unlike Proserin, stimulates the conduction of nerve impulses not only in the neuromuscular synapse, but also in the central nervous system, which explains the positive effect of this drug on mnestic processes.
Attention should be paid to the extremely important, although moderately pronounced property of Neuromidin - the ability to block sodium permeability of the membrane. This effect is associated with its sedative and analgesic properties. In addition, the drug has an antiarrhythmic effect.
Prozerin and Neuromidin are metabolized in the liver and are excreted mainly through renal mechanisms. 50% of Proserin and 3.7% of Neuromidin are released unchanged, which indicates the accelerated metabolism of Neuromidin. The initial effect from the administration of Proserin is observed, on average, after 10–15 minutes, and from the administration of Neuromidin - after 15–20 minutes. The duration of action of Neuromidin is 3–5 hours, which is associated with a long-term blockade of potassium permeability of the membrane, and Prozerin ends its effect 2–3 hours after its administration.
Prozerin, as a result of its effect on H-cholinergic receptors, leads to a significant increase in the force of contraction of skeletal muscles and is therefore indicated for use primarily in patients with myasthenia and myasthenic crisis. The drug is also used in neurological practice for paralysis, paresis that occurs after mechanical injuries, residual effects of meningitis, polio, encephalitis and neuropathies.
Due to its muscarinic-like effects, Prozerin is used to eliminate postoperative atony of the intestines and bladder, occasionally with weakness of labor, and sometimes in ophthalmological practice with open-angle glaucoma.
Due to the fact that Prozerin facilitates the transmission of excitation in the autonomic ganglia, it is indicated for poisoning with ganglion blockers. In addition, Prozerin is effective in case of overdose of muscle relaxants with antidepolarizing action (muscle weakness, respiratory depression).
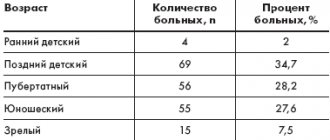
Indications for the use of Neuromidin are broader, since it is based on a biologically beneficial combination of two molecular effects (blockade of potassium permeability of the membrane and inhibition of cholinesterase) and it stimulates structural formations of the central nervous system. These are not only lesions of the peripheral nervous system: neuropathies, polyneuropathy, myelopolyradiculoneuritis, myasthenia gravis and myasthenic syndrome, multiple sclerosis and other forms of demyelinating diseases of the nervous system (in complex therapy), but also bulbar palsies and paresis with organic lesions of the central nervous system in the recovery period, as well as impairments of memory, attention, motivation and initiative of various origins, disorientation, emotional lability in senile dementia, Alzheimer's disease and other forms of late-life dementia, encephalopathy of traumatic, vascular and other origins, accompanied not only by memory impairment, but also by motor functions.
Neuromidin, like Proserin, is used for intestinal atony and intoxication with anticholinergic drugs.
A comparison of the clinical effect of Neuromidin and Proserin in myasthenia gravis showed that Proserin is significantly superior to Neuromidin in its anticholinesterase action. It is possible to achieve complete compensation of movement disorders in patients with myasthenia gravis only by using Proserin. However, its use is limited by a large number of side effects.
At the same time, the effectiveness of Neuromidin in the complex treatment of patients with myasthenia and myasthenic syndromes was noted, since it complements the action of Proserin. Both drugs improve the transmission of excitation at the neuromuscular synapse, and although Neuromidin is weaker in anticholesterase activity, it has a longer duration of action.
Neuromidin leads to a decrease in myasthenic crises, a faster onset and increase in the duration of remissions, an increase in muscle contractile activity, and an improvement in the general condition of patients.
According to the Russian Myasthenic Center, Neuromidin demonstrated maximum effectiveness in patients with Lambert-Eaton myasthenic syndrome, in contrast to Proserin, while a relative decrease in the severity of autonomic-trophic disorders was noted.
And yet, a reliable differential diagnostic test in the diagnosis of acute conditions in myasthenia gravis, namely myasthenic and cholinergic crises, is the administration of Proserin. A test with Proserin in a myasthenic crisis is positive and manifests itself in an increase in muscle strength or its complete restoration, and in a cholinergic crisis it is negative.
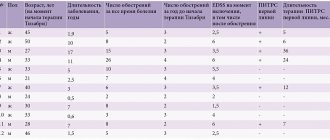
Clinical studies have shown the advantages of Neuromidin compared to Proserin in the complex treatment of facial nerve neuropathy, traumatic neuropathies and plexopathies. Treatment with Neuromidin can be started at the onset of the disease, and Proserin is prescribed 10 days later, taking into account the risk of post-paralytic contracture. Restoration of muscle function occurs faster when using Neuromidin. A clear positive effect was achieved in 89.5% of patients with damage to the facial muscles, in 80% of patients with traumatic neuropathies and plexopathies. Complete restoration of lost limb function was recorded in 40% of patients. Moreover, the activation of reinnervation of patients is confirmed by the positive dynamics of electromyography (EMG) indicators. It should be emphasized that there were no signs of contracture of the limbs or facial muscles in any of the observed patients. Despite the late administration of Proserin, several patients still experienced the appearance of pathological synkinesis on days 16–22 of the disease.
Neuromidin in the treatment of patients with demyelinating diseases of the nervous system (multiple sclerosis, amyotrophic lateral sclerosis) demonstrated a positive effect in the early stages of the disease in 40% of patients. Of course, the therapeutic effect of Neuromidin is less pronounced, but it is longer than that of Proserin. Under the influence of Neuromidin, the conduction of excitation in the spinal cord and peripheral nervous system is restored, which is manifested by a decrease in the severity of paresis and paralysis, sensitivity disorders, and an increase in strength in the affected muscle groups.
Effective treatment of sensory symptoms, regardless of the pathogenesis of peripheral nerve damage, is due to the blocking of ion channels of the axolemma and neuronal membranes, mainly potassium and partially sodium, which distinguishes Neuromidin from Proserin.
The analgesic effect of Neuromidin, which blocks ion channels, is achieved by inhibiting ectopic discharges and ephaptic transmission of excitation in the damaged nerve, in regenerating axonal branches as a result of collateral sprouting, as well as reducing the excitability of central neurons.
Neuromidin is part of a group of drugs used as a pathogenetic treatment of diabetic chronic distal symmetric sensorimotor polyneuropathy. The pathogenetic mechanisms of diabetic polyneuropathy lead to the progressive loss of myelinated fibers, as a result of which the conduction of the nerve impulse slows down, and in the “new” synapses newly formed as a result of regenerative sprouting, neuromuscular transmission is not effective. A study of Neuromidin in the treatment of diabetic polyneuropathy demonstrated a decrease in the severity of sensory symptoms, especially paresthesia, cramps, shooting pain, confirmed by EMG results (increased amplitude of the M-response).
Neuromidin, unlike Proserin, which does not actually penetrate the blood-brain barrier, has the ability to stimulate structural formations of the central nervous system, in particular cognitive processes.
Cognitive disorders are the leading clinical manifestations of chronic cerebrovascular insufficiency. There is an opinion that mnestic disorders are associated with central cholinergic deficiency, and the glutamate excitotoxicity that occurs in this case is secondary.
In older people, both the presynaptic and postsynaptic components of the central cholinergic synapses located in the cerebral cortex and hippocampus are impaired, resulting in decreased acetylcholine release and reduced sensitivity to it in old age.
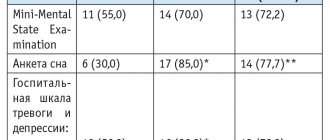
After a course of Neuromidin in patients with progressive vascular leukoencephalopathy, short-term memory improved, manifestations of asthenic syndrome decreased, and cognitive function indicators improved.
In patients with senile dementia of the Alzheimer's type and multi-infarct dementia who received Neuromidin, a decrease in behavioral disorders, improvement in cognitive functions, speech and social adaptation were detected.
An independent factor in cognitive impairment and the development of dementia is diabetes mellitus. Conducted clinical studies allow us to consider Neuromidin as a first-line drug for the treatment of patients with diabetes mellitus with moderate cognitive impairment and dementia.
In patients with the consequences of moderate or mild cerebral stroke, during treatment with Neuromidin, physical activity, intellectual functions, and memory capacity increased. During the recovery period of moderate ischemic stroke, patients experienced a significant decrease in the degree of paresis of the limbs without an increase in muscle tone, bulbar paralysis and paresis, regression of sensory disorders, and vegetative-trophic disorders. In 70–80% of patients with vascular and other organic lesions of the brain in the acute and long-term periods, there was a rapid regression of impairments in memory, attention, praxis, unsteadiness when walking, a decrease in asthenic phenomena, improved sleep, and an increase in the patient’s intellectual capabilities and performance. Almost all patients with aphasia showed improvement in speech functions.
Both drugs are produced in tablet form (Prozerin 15 mg, Neuromidin 20 mg) and ampoule form 1 ml (Prozerin 0.05%, 0.5% solution is used in ophthalmic practice, Neuromidin 1.5% and 0.5% solution ). Prozerin is also available in powder and granule form (for children). It is used orally and subcutaneously. For adults, Proserin is prescribed orally 30 minutes before meals in powders or tablets 10–15 mg 2–3 times a day, 0.5 mg (1 ml of 0.05% solution) is administered subcutaneously 1–2 times a day, into the eyes 1-2 drops of 0.5% solution 1-4 times a day.
With the development of myasthenic crisis in adults, Proserin is administered intravenously - 0.5–1 ml of a 0.05% solution, then subcutaneously in a dose of 1.5–2.5 ml, depending on the patient’s weight. Treatment of myasthenia gravis with Proserin is carried out for a long time. For other diseases, the course of treatment lasts 25–30 days; if necessary, it is repeated after a 3-4 week break.
Neuromidin is used orally, regardless of food intake, subcutaneously and intramuscularly. For lesions of the peripheral nervous system - 20 mg 2-3 times a day for 30-40 days. Courses of treatment are repeated with a break of 1–2 months. For acute neuritis, use 10–20 mg per day for 10–15 days; for chronic neuropathies, the duration of treatment is increased to 20–30 days. If necessary, courses are repeated 2-3 times with an interval of 2-4 weeks until maximum effect is achieved.
For myasthenia gravis, myasthenic syndromes, and demyelinating lesions of the nervous system, the dose of Neuromidin can be increased to 20–40 mg 5–6 times a day. The maximum daily dose is 180–200 mg. Relief of myasthenic crisis - 5-15 mg 1-2 times a day subcutaneously or intramuscularly.
The greatest effect for stimulating cognitive processes was obtained when using medium (50–60 mg/day) and high doses (80–120 mg/day) of Neuromidin.
For Alzheimer's disease and other forms of dementia, individual selection of the Neuromidin dosage regimen is required. The maximum daily dose can reach 200 mg; The duration of the course of therapy is from 4 months to 1 year. If it is necessary to use high doses, it is advisable to use 4-5 times a day with an interval of 2-3 hours.
In the recovery period of mild to moderate ischemic stroke, use a 1.5% solution of Neuromidin intramuscularly for 14 days, 1 ml per day.
For traumatic brain injury, Neuromidin is prescribed from 40 mg to 120–160 mg per day. The course of treatment is 30–40 days, 4 times a year with an interval of a month.
For the treatment and prevention of intestinal atony - 15–20 mg 2–3 times a day for 1–2 weeks.
Contraindications to the use of Proserin and Neuromidin are: hypersensitivity, epilepsy, extrapyramidal disorders with hyperkinesis, bronchial asthma, angina pectoris, severe bradycardia, pregnancy and lactation.
The number of contraindications and side effects for Proserin is greater compared to Neuromidin, which is associated with its strong anticholinesterase activity. Prozerin should not be used in cases of severe atherosclerosis, organic diseases of the heart and blood vessels, thyrotoxicosis, peptic ulcer of the stomach and duodenum, peritonitis, inflammatory processes in the abdominal cavity, mechanical obstruction of the gastrointestinal tract and urinary tract, prostate adenoma, period of acute illness, intoxication.
Caution should be exercised when using Neuromidin in patients with diseases of the cardiovascular system, thyrotoxicosis, and gastric ulcers, however, these conditions are not contraindications to its use.
When using Proserin, hypersalivation, profuse sweating, spastic contractions and increased intestinal motility, nausea, vomiting, flatulence, diarrhea, and frequent urination may occur. In addition, headache, dizziness, weakness, loss of consciousness, drowsiness, miosis, visual disturbances, arthralgia, allergic reactions including anaphylaxis may develop. From the cardiovascular system: slow heart rate, arrhythmias, AV block, nonspecific changes on the ECG, decreased blood pressure, cardiac arrest. From the respiratory system: shortness of breath, bronchospasm, respiratory depression to the point of stopping. Due to nicotine-like effects, Prozerin can cause tremors, spasms and twitching of skeletal muscles, including fasciculations of the tongue muscles, dysarthria, and convulsions.
To relieve side effects, reduce the dose of Proserin or stop taking it, and, if necessary, administer anticholinergic drugs.
When parenterally administering large doses of Prozerin, preliminary or simultaneous administration of atropine is necessary to neutralize the side effects of muscarinic. For developed convulsions, Sibazon is used.
Neuromidin, in comparison with Proserin, is well tolerated, side effects occur rarely, are mild and, as a rule, do not require discontinuation of the drug, due to weak anticholesterase activity and rapid elimination, which prevents accumulation and the occurrence of side effects.
In addition, when taking Neuromidin, unlike Proserin, there are no side effects caused by the effect on N-cholinergic synapses - fasciculations, cramps. Side effects of Neuromidin are associated with activation of M-cholinergic synapses. These are mainly dyspeptic symptoms (nausea, increased peristalsis, increased stool frequency without diarrhea), which regress on their own and do not require cessation of treatment.
The appearance of other side effects, such as dizziness, hypersalivation and bradycardia, skin allergic reactions, requires dose adjustment or a short 1-2-day break in treatment.
To prevent side effects, it is necessary to titrate the drug to a therapeutic dose so that the body adapts to increased cholinergic activity.
Due to the fact that Neuromidin can have a sedative effect, during treatment it is necessary to refrain from engaging in potentially hazardous activities that require increased concentration, including driving. During treatment, you should avoid taking ethanol, as adverse side effects may increase.
Neuromidin and proserin, despite the general anticholinesterase effect, are drugs with heterogeneous pharmacological properties, which determines their range of use. Due to its high anticholinesterase activity, Prozerin has a number of significant side effects and remains the drug of choice for myasthenia gravis, myasthenic crises and demyelinating diseases. Neuromidin has a wider spectrum of pharmacological activity, including a stimulating effect on the structural formations of the central nervous system, therefore it is successfully used both in the treatment of diseases of the peripheral nervous system and organic lesions of the central nervous system. The undoubted advantage is its good tolerability and the absence of pronounced side effects, which allows its long-term use in elderly patients suffering from numerous concomitant diseases.
For questions regarding literature, please contact the editor.
Z. A. Suslina , Doctor of Medical Sciences, Professor, Academician of the Russian Academy of Medical Sciences M. N. Sharov , Doctor of Medical Sciences, Professor O. A. Stepanchenko , Candidate of Medical Sciences MGMSU , Moscow