Instructions for use Ecoral 50 mg 50 pcs. soft capsules
Composition and release form
Capsules - 1 capsule:
- active substance: cyclosporine - 25 mg;
- excipients: ethyl alcohol - 39.9 mg; macrogol glyceryl hydroxystearate - 73.7 mg; polyglyceryl (3) oleate - 82.7 mg; polyglyceryl (10) oleate - 50.0 mg; D,L-alpha tocopherol - 0.25 mg;
- gelatin capsule shell: gelatin - 95.1 mg; glycerin 85% - 44.3 mg; sorbitol solution - 8.6 mg; iron oxide yellow - 0.1 mg; titanium dioxide - 0.8 mg; glycine - 1.1 mg.
Capsules - 1 capsule:
- active substance: cyclosporine - 50 mg;
- excipients: hydroxystearate - 147.4 mg; polyglyceryl (3) oleate - 165.3 mg; polyglyceryl (10) oleate - 100 mg; D,L-alpha tocopherol - 0.5 mg;
- gelatin capsule shell: gelatin - 222.2 mg; glycerin 85% - 103.8 mg; sorbitol solution - 20.2 mg; iron oxide yellow - 0.8 mg; titanium dioxide - 0.5 mg; glycine - 2.5 mg.
Capsules - 1 capsule.
- active substance: cyclosporine - 100 mg;
- excipients: ethyl alcohol - 159.6 mg; macrogol glyceryl hydroxystearate - 294.7 mg; polyglyceryl (3) oleate - 330.7 mg; polyglyceryl (10) oleate - 199.9 mg; D,L-alpha tocopherol - 1 mg;
- gelatin capsule shell: gelatin - 315.2 mg; glycerin 85% - 148.3 mg; sorbitol solution - 28.7 mg; iron oxide brown - 0.7 mg; titanium dioxide - 3.6 mg; glycine - 3.6 mg.
Contour cell packaging contains 10 pcs.; There are 5 packs in a cardboard pack.
Oral solution 100 mg/ml - 1 ml:
- active substance: cyclosporine - 100 mg;
- excipients: ethanol - 120 mg; macrogolglycerol hydroxystearate - 280 mg; polyglycerol (3) oleate - 310 mg; polyglycerol (10) oleate - 190 mg.
In a dark glass bottle - 50 ml; in a plastic case - 1 bottle.
Description of the dosage form
25 mg capsules: opaque, yellow, 12.5 x 8 mm, soft, oval gelatin capsules.
50 mg capsules: opaque, brown-yellow, 21x8 mm, oblong soft gelatin capsules.
100 mg capsules: opaque, brown, 26x8 mm, oblong soft gelatin capsules.
Each capsule strength is identified by a printed double triangle and text: “25 mg”, “50 mg”, “100 mg”.
The contents of the capsules are a transparent oily liquid from yellow to yellow-brown. Each capsule strength is indicated by IVAX printed and text: "25 mg", "50 mg" and "100 mg" respectively.
Oral solution 100 mg/ml: transparent yellowish to yellow-brown oily liquid.
Characteristic
It is a cyclic polypeptide consisting of 11 amino acids.
pharmachologic effect
At the cellular level, it inhibits the formation and release of lymphokines, including IL-2 (T-lymphocyte growth factor). Blocks resting lymphocytes in the G0 or G1 phase of the cell cycle and suppresses antigen-dependent release of lymphokines by activated T lymphocytes. All data obtained indicate that cyclosporine acts specifically and reversibly on lymphocytes.
Does not inhibit hematopoiesis and does not affect the function of phagocytic cells.
Pharmacokinetics
After oral administration, Cmax in the blood is observed in the range from 1 to 6 hours, with bioavailability averaging 30% (20–50%) and increasing with increasing dose and duration of treatment. Absorption decreases after liver transplantation, with liver disease or gastrointestinal pathology (diarrhea, vomiting, intestinal obstruction).
Intensively binds to proteins and formed elements of blood (concentration in whole blood is 2–9 times higher than in plasma). Protein binding - 90% (mainly with lipoproteins). Distributed mainly outside the bloodstream: 33–47% is present in plasma, 4–9% in lymphocytes, 5–12% in granulocytes, 41–58% in erythrocytes. After oral administration, Tmax in plasma is 1.5–3.5 hours. It is intensively metabolized in the liver by cytochrome P4503A, to a lesser extent in the gastrointestinal tract and kidneys with the formation of 15 identified metabolites. Excreted in bile; kidneys with urine - 6% of the dose administered orally. Excreted in breast milk.
T1/2 in adults - 19 hours, in children - 7 hours, regardless of dose or route of administration.
Indications for use
Graft-versus-host disease (GVHD) - prevention and treatment; autoimmune diseases.
Indications related to transplantation: solid organ transplantation (prevention of graft rejection after allogeneic transplantation of kidney, liver, heart, combined heart-lung, lung or pancreas; treatment of graft rejection in patients previously treated with other immunosuppressants; bone marrow transplantation (prevention of rejection graft after bone marrow transplantation, prevention and treatment of GVHD.
Indications not related to transplantation: endogenous uveitis (active, sight-threatening uveitis of the middle and posterior part of the eye of non-infectious etiology, if conventional therapy is unsuccessful or leads to severe adverse reactions), Behcet's uveitis with recurrent attacks of inflammation affecting the retina; nephrotic syndrome (steroid-dependent and steroid-resistant forms in remission); rheumatoid arthritis (severe forms), psoriasis (severe forms when systemic therapy is required).
Contraindications for use
Hypersensitivity to the components of the drug, malignant neoplasms and precancerous skin diseases, chicken pox, Herpes zoster (risk of generalization of the process), severe liver failure, hyperkalemia, hypertension, malabsorption syndrome, infectious diseases in the acute phase.
Use during pregnancy and children
Experience with the use of cyclosporine in pregnant women is limited. Data obtained from patients in the post-transplant period show that treatment with cyclosporine increases the risk of negative effects on the course and outcome of pregnancy. If it is necessary to prescribe Ecoral, breastfeeding should be stopped.
Price for Ecoral in Moscow Buy Ecoral at REDapteka.ru
Instructions for use for Ecoral.
Side effects
In the first week of using the drug, you may experience a burning sensation on the skin of the extremities. After organ transplantation, the most common occurrences are: hypertrichosis, tremor, renal dysfunction and liver dysfunction, gum hypertrophy, gastrointestinal disorders (anorexia, nausea, vomiting); dose-dependent and reversible increase in the level of creatinine, urea, bilirubin, liver enzymes in the blood serum (careful monitoring of parameters and adjustment of the dosage of cyclosporine is necessary).
Hypertension most often develops during heart transplantation; this side effect occurs less frequently during kidney transplantation.
The most common symptoms that occur during bone marrow transplantation are: gastrointestinal disorders, tremor, hypertrichosis, and possible facial swelling. Children experience swelling and cramps.
Adverse reactions are moderate and are usually eliminated by reducing the dosage of the drug.
Drug interactions
Inducers or inhibitors of cytochrome P450 can reduce or increase the concentration of cyclosporine in the blood.
Drugs that reduce the concentration of cyclosporine: barbiturates, carbamazepine, phenytoin, nafcillin, sulfadimidine (with intravenous administration); rifampicin, octreotide, probucol, orlistat; preparations containing St. John's wort (Hypericum perforatum); troglitazone.
Drugs that increase the concentration of cyclosporine: some antibiotics - macrolides (mainly erythromycin and clarithromycin); ketoconazole, fluconazole, itraconazole, diltiazem, nicardipine, verapamil, metoclopramide, oral contraceptives, danazol, methylprednisolone (high doses); allopurinol, amiodarone, cholic acid and its derivatives.
Oral erythromycin should be avoided (increases the concentration of cyclosporine in the blood). If, due to the lack of alternative therapy, erythromycin is prescribed, it is recommended to carefully monitor the concentration of cyclosporine in the blood, renal function and the presence of side effects of cyclosporine.
Caution should be exercised when prescribing drugs with nephrotoxic effects simultaneously, for example: aminoglycosides (including gentamicin, tobramycin), amphotericin B, ciprofloxacin, vancomycin, trimethoprim (+ sulfamethoxazole), NSAIDs (including diclofenac, naproxen, sulindac ), melphalan.
Vaccinations may be less effective during treatment with cyclosporine; The use of live attenuated vaccines should be avoided.
Combined use with nifedipine may lead to more pronounced gingival hyperplasia than with cyclosporine monotherapy.
May significantly increase the bioavailability of diclofenac (probably due to decreased metabolism) with the possible development of reversible renal impairment.
May reduce the clearance of digoxin, colchicine, lovastatin and prednisolone, leading to increased toxic effects, in particular muscle pain, weakness, myositis and, in rare cases, rhabdomyolysis.
Dosage
Inside. Capsules should be swallowed whole with water. The daily dose is divided into 2 doses. The drug dosage ranges below are guidelines only. The concentration of cyclosporine in the blood should be monitored. Based on the results obtained, the dose required to achieve the desired level of cyclosporine concentration in various patients is determined.
Transplantation
For solid organ transplantation, the drug is prescribed 12 hours before surgery at a dose of 10–15 mg/kg/day, divided into 2 doses. For 1-2 weeks after surgery, the drug is prescribed daily at the same dose, then, under the control of the concentration of cyclosporine in the blood, it is gradually reduced until a maintenance dose is reached - 2-6 mg/kg/day in 2 doses.
In cases where Ecoral is prescribed in combination with glucocorticoids, as well as as part of three-component or four-component therapy, the dose of the drug can be reduced even at the initial stage of therapy (3-6 mg/kg/day in 2 doses) or adjusted during treatment with taking into account the concentration of cyclosporine in the blood and the dynamics of safety indicators (concentration of urea, serum creatinine and blood pressure).
For bone marrow transplantation, the initial dose should be prescribed one day before surgery. The daily dose is 12.5 mg/kg in 2 divided doses. Maintenance therapy is carried out for at least 3 months (preferably 6 months), after which the dose of cyclosporine is gradually reduced over the course of a year.
Indications not related to transplantation
Endogenous uveitis: to achieve remission, the initial daily dose is 5 mg/kg in 2 divided doses until signs of inflammation disappear and visual acuity improves. In cases that are difficult to treat, the dose can be increased for a short time to 7 mg/kg/day. If Ecoral monotherapy is ineffective, systemic glucocorticoids are added to complex treatment in a daily dose of 0.2–0.4 mg/kg prednisolone (or another glucocorticoid drug in an equivalent dose). Once the clinical effect is achieved, the dose of Ecoral is gradually slowly reduced to the lowest effective dose, which during the period of remission of the disease should not exceed 5 mg/kg/day.
Nephrotic syndrome: to achieve remission, the recommended daily dose is 5 mg/kg for adults and 6 mg/kg for children (in 2 doses), provided that renal function is normal. In case of impaired renal function, the initial dose should not exceed 2.5 mg/kg/day. If Ecoral monotherapy is ineffective, especially in steroid-resistant patients, it is recommended to combine the drug with oral glucocorticoids in low doses. If there is no clinical effect within 3 months, Ecoral should be discontinued.
Rheumatoid arthritis: during the first 6 weeks of treatment, the recommended dose is 3 mg/kg/day in 2 divided doses. In case of ineffectiveness and if tolerance allows, the daily dose can be gradually increased, but not more than 5 mg/kg. For maintenance therapy, the dose is selected individually depending on the tolerability of the drug. Ecoral can be combined with low doses of glucocorticoids and/or NSAIDs, with a weekly course of methotrexate in low doses in patients with an unsatisfactory response to monotherapy with the latter. The initial dose of Ecoral is 2.5 mg/kg/day (in 2 doses), the dose can be increased to a level limited by tolerability.
Psoriasis: treatment is selected individually, the recommended initial dose for inducing remission is 2.5 mg/kg/day in 2 divided doses. If there is no improvement within 1 month of treatment, the daily dose can be increased, but not more than 5 mg/kg. If after 6 weeks therapy with a dose of 5 mg/kg/day is unsuccessful or the effective dose does not meet established safety parameters, the drug should be discontinued. The use of an initial dose of 5 mg/kg/day is justified in patients whose condition requires rapid improvement. If a satisfactory effect is achieved, then Ecoral can be discontinued, and a subsequent relapse can be treated by re-prescribing this drug at the previous effective dose. Some patients may require long-term maintenance therapy (the dose is selected individually at the minimum effective level and should not exceed 5 mg/kg/day).
Atopic dermatitis: The recommended initial dose range is 2.5–5 mg/kg/day in 2 divided doses. If the initial dose of 2.5 mg/kg/day does not achieve a satisfactory response within 2 weeks, the daily dose can be quickly increased to a maximum of 5 mcg/kg. In very severe cases, a rapid and adequate effect in treating the disease can be achieved using an initial dose of 5 mg/kg/day. Once a satisfactory response is achieved, the dose is gradually reduced and, if possible, the drug is discontinued. If a relapse occurs, a repeat course is given. Although an 8-week course of treatment may be sufficient to clear the skin, therapy for up to 1 year has been shown to be effective and well tolerated, subject to mandatory monitoring of all necessary indicators.
Overdose
There are no data on overdose with the drug to date and there is limited experience with overdose of other cyclosporines.
Symptoms: impaired renal function, which is probably reversible and disappears after discontinuation of the drug.
Treatment: according to indications - general supportive measures. The drug can only be eliminated from the body using nonspecific measures, including gastric lavage, since hemodialysis and hemoperfusion using activated charcoal are ineffective.
On the issue of the safety of prescribing Ecoral in patients after cadaveric kidney allotransplantation
The article presents the results of a clinical study of the comparative bioequivalence of original (Neoral) and generic (Ekoral) drugs in patients with a transplanted kidney. It was found that the drug Ecoral can be considered bioequivalent to the original cyclosporine, since it meets the required safety parameters for generic drugs with a narrow therapeutic range.
Rice. 1. Average pharmacokinetic profiles of the drugs Neoral and Ecoral
Rice. 2. Diagram of average values and 90% confidence intervals of AUCtr for Neoral and Ecoral
Rice. 3. Diagram of mean Cmax values and 90% confidence intervals for Neoral and Ecoral
Rice. 4. Pharmacokinetic curves for patients in whom Cmax was reached 1.5 hours after taking the drugs
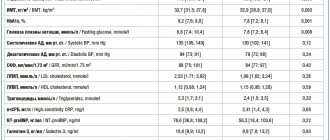
Table 1. Values of the main pharmacokinetic parameters of the drugs Neoral and Ecoral
Table 2. Dynamics of cyclosporine concentration in the study of Neoral and Ecoral, indicating the significance coefficient of the differences
Table 3. Average concentrations of cyclosporine in the group of patients in whom Cmax was reached one and a half hours after taking the drugs
Introduction
The biological effects of cyclosporine and its selective effect on T cells and its inhibitory effect on the production of interleukin-2 were discovered by JF Borel et al. in the 1970s [1]. In 1982, cyclosporine was introduced into the immunosuppression protocol for human organ transplantation. Since 1995, the original form of cyclosporine, oil-based Sandimmune, has been largely replaced by the microemulsion Neoral, due to its higher bioavailability compared to Sandimmune [2]. After the expiration of the patent term (20 years) of the original drug, any pharmaceutical company has the right to reproduce a generic cyclosporine. Thus, at the beginning of 2006, more than six analogues of cyclosporine from domestic and foreign manufacturers were registered in the Russian Federation. At that time, pharmacokinetic studies of two generic analogues of cyclosporine were carried out in our clinic [3].
The main requirement for a generic drug is confirmation of its bioequivalence (“pharmacokinetic equivalence”), that is, the drug must have comparable bioavailability when studied under similar experimental conditions [4, 5]. The importance of a serious and comprehensive approach to studying the equivalence of generic cyclosporine is evidenced by the fact that the generic cyclosporine Sang Cya (SangStat Medical Corporation of Fremont, USA) in the form of an oral solution, which passed all tests and was registered in the USA in 1997, was later withdrawn from the market due to lack of bioequivalence, as additional studies showed that its absorption was sharply reduced when taken concomitantly with apple juice [6].
The main feature of the clinical use of cyclosporine is that it belongs to the group of “critical dose drugs”, which implies a narrow therapeutic range of the drug, when the therapeutic concentration is close to toxic [5]. In other words, underdosing entails an insufficient immunosuppressive effect and, as a consequence, transplant rejection [3, 7], and conversely, even a small increase in drug concentration is dangerous due to the possibility of nephrotoxicity or other undesirable reactions [8, 9]. That is why, for generic drugs with a narrow therapeutic range, the limits of compliance with the original drug are narrowed from 80–125% to 90–111% [5]. Thus, the mean values and their 90% confidence interval (CI) for the generic drug under study must correspond precisely to these limits of the corresponding values for the original drug. Only in this case can the test cyclosporine preparation be considered bioequivalent to the original one.
There is another feature of cyclosporine - inter- and intra-individual variability of pharmacokinetic parameters, that is, differences in bioavailability are possible not only in different patients, but even in each individual patient [10, 11].
We conducted a study to assess the bioequivalence of the generic drug to the original drug in patients with a transplanted kidney, which would allow us to further judge the safety of transferring patients from Neoral (Novartis Pharma) to Ecoral (Ivex). To accomplish this task, we carried out monitoring based on determining the level of the drug in the blood for 12 hours after taking it. We compared the area under the concentration-time pharmacokinetic curve (AreaUnderCurve, AUC), which reflects the total amount of drug entering the body, in the same patients when taking Neoral and Ecoral. In addition, the highest cyclosporine concentrations (Cmax) at time Tmax were assessed.
Materials and methods
Pharmacokinetic studies were conducted in 42 patients (aged 23 to 67 years, mean age 51 ± 9 years) with allogeneic cadaveric kidney transplant (ALT). All patients are observed at the Moscow City Nephrology Center. The duration of transplantation at the time of examination ranged from 1 to 15 years (the average period after ATTP was 76 ± 50 months). The vast majority of patients had consistently satisfactory graft function. Only 4 (9.5%) recipients had graft dysfunction, but their creatinine levels were in the range of 150–190 μmol/L. All patients initially took Neoral. After a pharmacokinetic study, a conversion was made to Ecoral while maintaining the same dose of cyclosporine. After 14 days of taking Ecoral, a full pharmacokinetic study was repeated, with determination of concentration on an empty stomach, then after 1; 1.5; 2; 3; 4; 6; 8, 10 and 12 hours after taking cyclosporine per os. Along with this, azotemic blood parameters, levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), total cholesterol were re-determined and a general blood test was performed. Three patients stopped the study early due to the development of an adverse event, which was reported to the Federal State Institution “Scientific Center for Expertise of Medical Products” of Roszdravnadzor. There were no cases of exacerbation of gastrointestinal diseases or diarrhea that could alter the pharmacokinetics of cyclosporine.
The concentration of cyclosporine was determined by enzyme immunoassay (DimensionRXL, Siemens). For laboratory examination, an automatic biochemical analyzer Hitachi-902 (Roche) was also used, and the parameters of a general blood test were determined using an ABX Pentra XL 80 device (Horiba medical). The area under the pharmacokinetic curve (AUC) was calculated using the trapezoidal rule (AUCtr) and the Gaspari formula [12]:
AUC (Co + Q + С₃) = 5.189 × С₀ + + 1.267'Q + 4.15 × С₃ + 135.1.
Statistical processing of the material was performed using SPSS 11.5 for Windows. To test the hypothesis of normal distribution, the Kolmogorov–Smirnov test was used. Since this test established that the data obtained were subject to a normal distribution, we used a t-test for dependent samples to compare the corresponding indicators. The descriptive statistics method included the calculation of means with standard deviations. The results were considered statistically significant at p values
Research results
We initially compared the area under the pharmacokinetic curve for the two study drugs (Fig. 1). It was found that AUC values, both calculated using the trapezoidal rule and the Gaspari formula, do not have a statistically significant difference (Table 1). Thus, the area under the pharmacokinetic curve for Ecoral is 4204 ± 908 and is almost equal to the AUC of Neoral 4265 ± 902 at p = 0.6 (Fig. 1). The absorption profiles of Ecoral and Neoral (the area under the concentration-time curve during the first four hours) after taking the drugs also did not differ significantly and were equal to 2556 ± 596 when taking Neoral and 2457 ± 565 when taking Ecoral. This difference turned out to be insignificant, despite the fact that Neoral is a microemulsion and Ecoral is a microdispersion and they have different particle sizes of the drug substance.
To assess bioequivalence, in addition to analyzing graphs of pharmacokinetic curves of cyclosporine preparations, we needed to compare 90% confidence intervals of the found mean AUC values. For Neoral the 90% CI was in the range of 4021–4508, and for Ecoral the range was 3958–4450. The diagram (Fig. 2) shows that the 90% confidence interval for the average AUC value of the drug Ecoral is within the bioequivalence limits of Neoral (the appropriate level is from 3838 to 4734), that is, it corresponds to the permissible limit of 90–111% for drugs with a narrow therapeutic window.
The next step in assessing the bioequivalence of Ecoral to Neoral is the analysis of average Cmax values during the time period Tmax. In both cases, the concentration of cyclosporine was maximum one and a half hours after administration, while the concentration of cyclosporine after taking Neoral was slightly higher than the same indicator when taking Ecoral. However, these differences (concentration values of 1036 ± 309 ng/ml when taking Neoral and 931 ± 322 ng/ml when taking Ecoral) did not reach significant significance (p = 0.06). That is why it was very important to evaluate the peak concentrations (Cmax) of both drugs.
When assessing the mean Cmax values and their 90% confidence intervals, we obtained the following values: Cmax for Neoral was 1135 ± 254 ng/ml (90% CI 1066–1203), and for Ecoral – 1110 ± 278 ng/ml (90% CI 1035–1185).
Proof of bioequivalence is possible if the 90% confidence interval for Ecoral's Cmax is within the range of 1021–1260 ng/ml. Figure 3 shows that the 90% confidence interval for the mean Cmax of Ecoral is within the specified bioequivalence limits.
To compare drug absorption, we identified a group of patients (n = 18) from the total number of those examined with Cmax at point T1.5 when taking Neoral and compared their pharmacokinetic curves after taking Ecoral (Fig. 4). The choice of this particular point of the pharmacokinetic curve was not accidental: the maximum difference in cyclosporine concentrations was observed exactly one and a half hours after taking Neoral and Ecoral (Table 2). In almost half of the patients (44%), the time of maximum absorption of the drug shifted, which is explained by the intra-individual variability of cyclosporine.
By the fourth hour after taking Neoral, the concentration of cyclosporine in the blood of this group of patients decreased by 73% and amounted to 322 ± 107 ng/ml, and after taking Ecoral the concentration changed by 58% and amounted to 412 ± 234 ng/ml. No statistically significant differences were obtained using the t-test (p = 0.08). But statistically significant differences in the concentration values are shown at point C3, when the concentrations were 473 ± 152 ng/ml when taking Neoral and 568 ± 204 ng/ml when taking Ecoral (p = 0.014). By the 6th hour, the concentrations were equal and did not differ significantly until the 12th hour point (Fig. 3). AUC when taking Neoral was 4133 ± 776, and when taking Ecoral – 4289 ± 1019 (p = 0.46).
When evaluating generics, the absorption coefficient (ratio Cmax/AUCtr), which characterizes the rate of absorption of the drug, is also analyzed. When taking Neoral it was 0.27 ± 0.05, and when taking Ecoral it was 0.26 ± 0.04. The t-test carried out to compare these indicators did not establish a significant difference in the coefficients (p = 0.6).
In all patients, the indicators of general and biochemical blood tests at the end of the study, as well as at the beginning of the study, remained within normal limits. The average level of creatinine was 114 ± 26 µmol/l, urea – 10.8 ± 3.5 mmol/l, ALT – 24 ± 15 U, AST – 22 ± 9 U, total cholesterol – 5 ± 1.1 mmol/l, hemoglobin – 128 ± 16.7 g/l, leukocyte level – 7.5 ± 2.3 × 109/l.
Discussion of the results obtained
A fundamental issue when assessing the bioequivalence of drugs is to determine the area under the pharmacokinetic curve, which reflects the total amount of the substance entering the body and its maximum serum concentration Cmax. Our obtained average values and 90% confidence intervals of AUCtr and Cmax of the tested drug Ecoral confirm its bioequivalence to Neoral. At the same time, Ecoral meets the required safety parameters for generic drugs with a narrow therapeutic range. The Tmax indicator in both cases was at the one and a half hour point.
At the same time, we received data that indicate some difference in the absorption of the drug, although their absorption coefficients did not differ. The results of the analysis in the group of patients with a peak concentration at the one and a half hour point still indicate a different absorption profile.
After the Sandimmune Neoral microemulsion was created, the particle size of the active substance in the drug began to be considered as the most important factor affecting the bioavailability of cyclosporine [11]. To assess the effect of particle size on bioavailability, a comparison was made of the average area under the concentration-time curve for drugs that differ only in particle size [13]. It turned out that the bioavailability of dosage forms with maximum and minimum particle sizes did not have statistically significant differences. According to researchers, the reason for the high bioavailability of some dosage forms that form large particles in solution is the adhesive properties of such particles, which are capable of providing a large effective contact surface on the epithelial layer. It is likely that dispersed Ecoral particles are able to remain in contact with the wall of the gastrointestinal tract longer due to their bioadhesiveness.
Conclusion
According to our results, the drug Ecoral can be considered bioequivalent to Neoral, since it provides similar bioavailability to cyclosporine. Conversion to Ecoral in the kidney transplanted patients we examined was carried out with great caution. The importance of monitoring cyclosporine concentrations after transferring a patient to a generic drug should be emphasized. The transplantologist should inform the patient about the need to monitor the concentration of cyclosporine, blood creatinine and blood pressure levels after such a transition. The patient must be aware that it is prohibited to mix different dosage forms.
To date, there are insufficient studies examining the results of treatment with generic drugs in certain groups of patients. Thus, when switching from Neoral to generic Gengraf, a clinically significant change in the pharmacokinetics of sirolimus was identified in patients taking a combination of Gengraf and sirolimus. When taking this generic cyclosporine, the area under the pharmacokinetic curve of sirolimus was significantly lower than when taking Neoral [14]. This example demonstrates the importance of the moment of switching from the original drug to generic analogues and the need for close monitoring of the patient in each individual case.
The increase in the number of patients after allogeneic kidney transplantation, associated with improved graft survival rates, leads to increased healthcare costs, so the emergence of an increasing number of generic immunosuppressive drugs is quite predictable. Undoubtedly, the most important issue in their selection and evaluation should remain safety criteria for patients.